- Maxwell’s equations and the experiments of Heinrich Hertz in 1887 proved that light behaves as an electromagnetic wave.
- Experiments with electric discharge through gases at very low pressure helped scientists discover new particles and radiations.
- Wilhelm Conrad Roentgen discovered X-rays in 1895.
- J. J. Thomson discovered the electron in 1897.
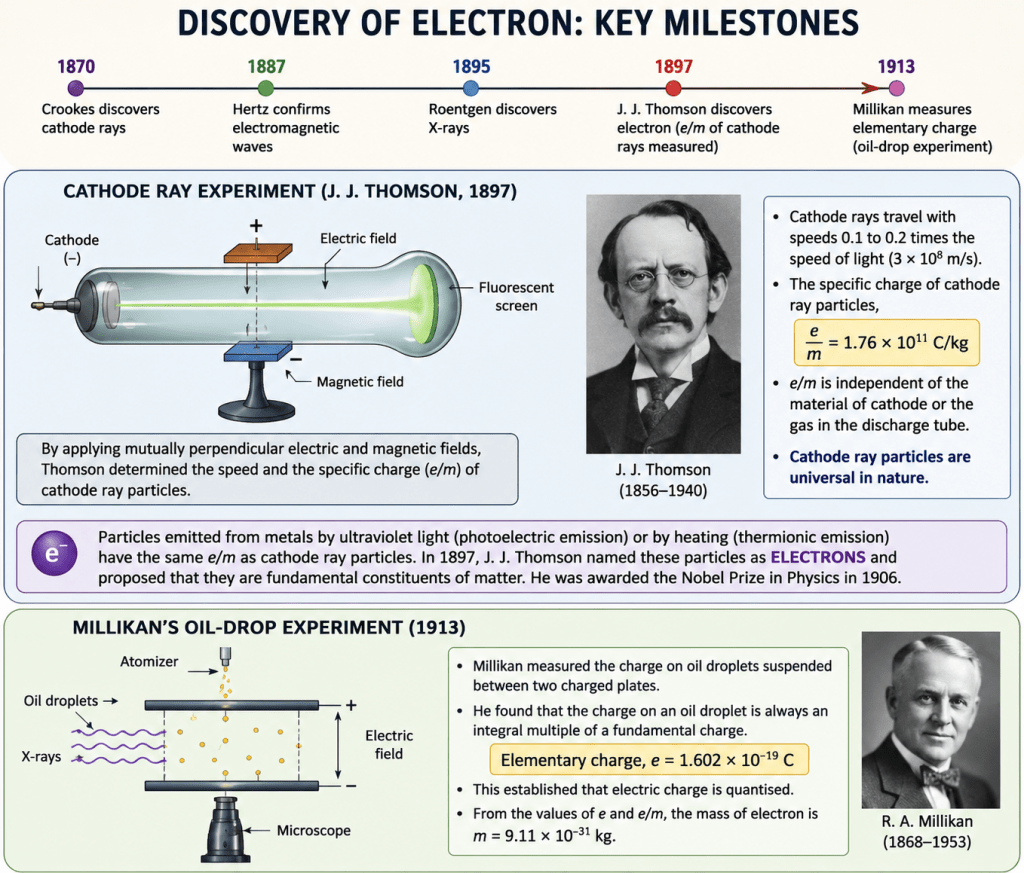
- In a discharge tube containing gas at very low pressure, applying a high electric field caused a discharge and produced a fluorescent glow on the glass wall opposite the cathode.
- William Crookes discovered cathode rays in 1870 and proposed that they were streams of fast-moving negatively charged particles.
- J. J. Thomson later confirmed this idea experimentally using electric and magnetic fields and measured the speed and charge-to-mass ratio of these particles, which were later called electrons.
This passage continues the story of the discovery of the electron and explains important experimental results.
Main points
- J. J. Thomson measured the specific charge of cathode ray particles:
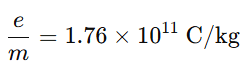
- The particles moved at speeds about .1C to .2C
where (c) is the speed of light.
- The value of (e/m) was the same regardless of:
- the metal used as the cathode,
- or the gas inside the discharge tube.
This showed that cathode ray particles are universal and present in all matter.
Discovery of electrons from other experiments
Scientists also found that:
- ultraviolet light falling on some metals caused emission of negative particles (photoelectric effect),
- heated metals also emitted negative particles (thermionic emission).
The emitted particles had the same (e/m) ratio as cathode rays, proving they were identical.
Naming of the electron
In 1897, J. J. Thomson named these particles electrons and proposed that they are fundamental constituents of matter.
He received the Nobel Prize in Physics in 1906 for this work.
Millikan’s oil-drop experiment
In 1913, Robert Andrews Millikan measured the charge of the electron using the famous oil-drop experiment.
He found the elementary charge:
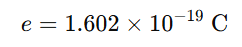
He also showed that electric charge is quantized, meaning charge always exists in whole-number multiples of this elementary charge.