Salt analysis is a systematic method used to detect the cation and anion present in a given salt using dry and wet tests.
METHODS OF SALT ANALYSIS
1. Dry Tests
- Flame test
- Borax bead test
- Charcoal cavity test
- Cobalt nitrate test
- Heating test
2. Wet Tests
- Preliminary test (solubility test)
- Anion analysis (acid radical tests)
- Cation analysis (group analysis)
- Confirmatory tests
3. Special Tests
- Brown ring test
- Lime water test
- Chromyl chloride test
- Lead acetate test
- Nessler’s test
CATION GROUPS (Basic Radicals)
Group I (Dilute HCl group)
- Ag⁺
- Pb²⁺
- Hg₂²⁺
Group II (H₂S in acidic medium)
Group II A (Copper group)
- Cu²⁺
- Cd²⁺
- Hg²⁺
- Pb²⁺
- Bi³⁺
Group II B (Arsenic group)
- As³⁺ / As⁵⁺
- Sb³⁺ / Sb⁵⁺
- Sn²⁺ / Sn⁴⁺
Group III (NH₄Cl + NH₄OH group)
- Fe³⁺
- Al³⁺
- Cr³⁺
Group IV (H₂S in basic medium)
- Zn²⁺
- Mn²⁺
- Ni²⁺
- Co²⁺
Group V ((NH₄)₂CO₃ group)
- Ba²⁺
- Sr²⁺
- Ca²⁺
Group VI (No group reagent)
- Mg²⁺
- Na⁺
- K⁺
- NH₄⁺
What is a Group Reagent?
A group reagent is a chemical reagent used in salt analysis to precipitate a specific group of cations together based on their similar properties.
Do anions have groups in salt analysis?
No, anions do not have a fixed “group system” like cations.
Why not?
- Cations are grouped based on systematic precipitation using group reagents
- Anions do not show such uniform precipitation behavior
- Different anions require different specific tests
In qualitative analysis, dry tests are performed without dissolving the salt in solution. In a flame test, the salt (usually converted to its chloride) is directly heated in a flame, so it falls under dry tests. Flame test is done by heating a salt (on a clean wire loop with HCl) in a non-luminous flame and observing the characteristic color.
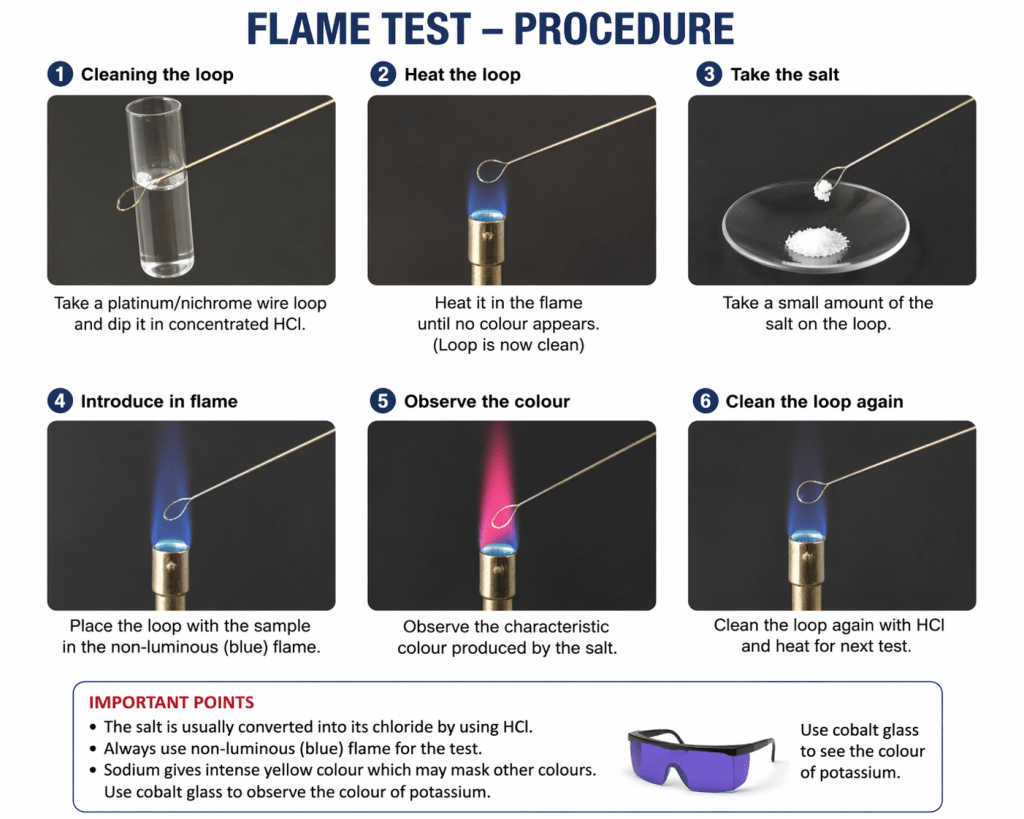
COMPLETE FLAME TEST LIST
1. Alkali Metals (Group 1) — Very important
- Li⁺ (Lithium) → Crimson red
- Na⁺ (Sodium) → Intense golden yellow
- K⁺ (Potassium) → Lilac / pale violet (seen through cobalt glass)
- Rb⁺ (Rubidium) → Red-violet
- Cs⁺ (Caesium) → Blue-violet
2. Alkaline Earth Metals (Group 2)
- Be²⁺ (Beryllium) → No colour
- Mg²⁺ (Magnesium) → No colour
- Ca²⁺ (Calcium) → Brick red (orange-red)
- Sr²⁺ (Strontium) → Crimson red
- Ba²⁺ (Barium) → Apple green
3. Transition & Post-Transition Metals
- Cu⁺ / Cu²⁺ (Copper) → Bluish green
- Pb²⁺ (Lead) → Bluish white (faint)
- Tl⁺ (Thallium) → Bright green
Most others do NOT give a characteristic flame colour:
- Fe²⁺ / Fe³⁺ → No colour
- Co²⁺ → No colour
- Ni²⁺ → No colour
- Zn²⁺ → No colour
- Mn²⁺ → No colour
- Al³⁺ → No colour
4. Ions Showing NO Flame Colour (Important for elimination)
- Be²⁺, Mg²⁺, Al³⁺
- Zn²⁺, Fe²⁺/Fe³⁺, Co²⁺, Ni²⁺, Mn²⁺
Reason: emission not in visible region / weak excitation
IMPORTANT EXAM FACTS
1. Sodium Interference
- Na⁺ gives very intense yellow
- Masks all other colours
Solution: Use cobalt glass to detect K⁺
2. Why flame colour appears?
- Due to electronic excitation and emission spectrum
3. Best observed for:
- Volatile salts (usually chlorides)
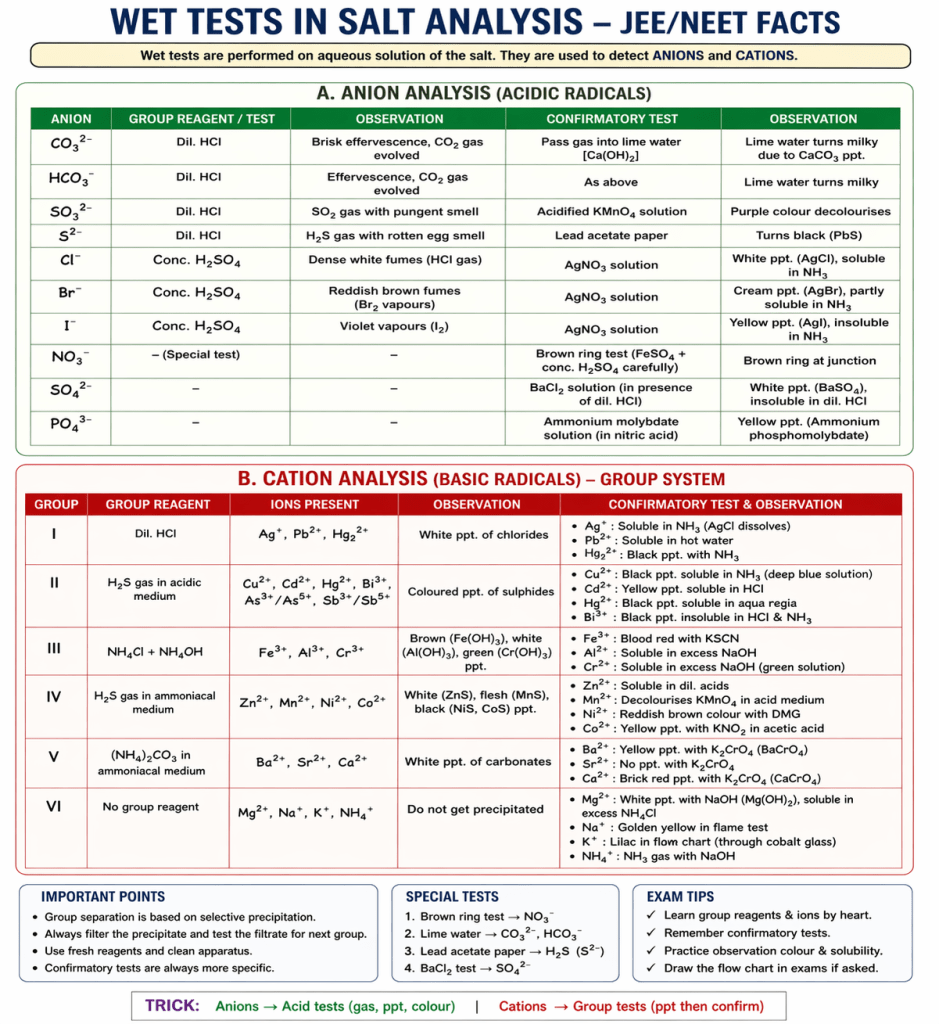
What is a Wet Test?
A wet test is a method in salt analysis where the substance is first dissolved in a solvent (usually water or acid) and then tested using chemical reactions.
COLOURS OF CATION RADICALS
1. Coloured Ions (VERY IMPORTANT)
- Cu²⁺ (Copper) → Blue solution
- Fe²⁺ (Ferrous) → Pale green
- Fe³⁺ (Ferric) → Yellow / brown
- Ni²⁺ (Nickel) → Green
- Co²⁺ (Cobalt) → Pink
- Cr³⁺ (Chromium) → Green / violet
- Mn²⁺ (Manganese) → Very pale pink
- V³⁺ (Vanadium) → Green
- Ti³⁺ (Titanium) → Purple
2. Colourless Ions (VERY COMMON MCQ)
- Na⁺
- K⁺
- NH₄⁺
- Mg²⁺
- Ca²⁺
- Ba²⁺
- Sr²⁺
- Al³⁺
- Zn²⁺
3. Characteristic Precipitate Colours (IMPORTANT)
- Ag⁺ → White ppt (AgCl)
- Pb²⁺ → White ppt
- Fe³⁺ → Reddish-brown ppt (Fe(OH)₃)
- Fe²⁺ → Dirty green ppt (Fe(OH)₂)
- Cu²⁺ → Blue ppt (Cu(OH)₂)
- Ni²⁺ → Green ppt
- Co²⁺ → Blue ppt (on heating)
- Zn²⁺ → White ppt (Zn(OH)₂)
- Al³⁺ → White gelatinous ppt
IMPORTANT EXAM POINTS
- Colour is due to d–d transitions (transition metals)
- d⁰ and d¹⁰ ions → colourless (Zn²⁺, Al³⁺ etc.)
- Cu²⁺ always → blue solution (very important)
MOST ASKED
- Blue solution → Cu²⁺
- Green solution → Ni²⁺ / Fe²⁺ (trap!)
- Pink → Co²⁺
- Yellow/brown → Fe³⁺
SPECIAL REAGENTS – DETAILS
1. Nessler’s Reagent
Used for: Detection of NH₄⁺ (ammonium ion)
Composition
- Alkaline solution of K₂HgI₄ (potassium mercuric iodide)
Observation
- Brown colour / brown precipitate → NH₄⁺ present
Reaction idea
- NH₃ (from NH₄⁺) reacts with Nessler’s reagent → coloured complex
One-line (viva)
Nessler’s reagent gives brown colour with ammonium ions.
2. FeSO₄ (Ferrous sulphate) – Brown Ring Test
Used for: Detection of NO₃⁻ (nitrate ion)
Reagents used
- Fresh FeSO₄ solution
- Concentrated H₂SO₄
Observation
- Brown ring at junction of two layers
Key concept
- Formation of nitrosyl complex
Important conditions
- FeSO₄ must be fresh
- H₂SO₄ added slowly along test tube wall
One-line (viva)
Brown ring test confirms presence of nitrate ion.
3. Copper Turnings Test
Used for: Detection of NO₃⁻ (nitrate ion)
Reagents used
- Copper metal + conc. H₂SO₄
Observation
- Brown fumes of NO₂ gas
Reaction idea
- Nitrate gets reduced → NO₂ gas evolves
TEST FOR NICKEL (Ni²⁺) – KEY FACTS
1. Group Information
- Group IV cation
- Precipitated as NiS (black ppt) in basic medium (H₂S + NH₄OH)
2. Most Important Confirmatory Test
Dimethylglyoxime (DMG) Test
- Reagent: Dimethylglyoxime (DMG) + NH₄OH
- Observation: Bright red / rose-red precipitate
- Compound formed: Nickel dimethylglyoxime complex
Very specific test for Ni²⁺
3. Reaction Idea
- Ni²⁺ + DMG → Red complex (insoluble)
4. Other Supporting Facts
- Ni²⁺ solution → Green colour
- With NaOH → Green ppt (Ni(OH)₂)
- With NH₄OH → forms complex (no ppt in excess)
What is Lake Test?
Lake test is used to detect Al³⁺ (aluminium ion) in salt analysis.
Principle
- A lake is a coloured complex (dye + metal hydroxide) formed when a dye gets adsorbed on a gelatinous precipitate.
Procedure (short)
- Add NH₄OH → forms Al(OH)₃ (white gelatinous ppt)
- Add a dye (like alizarin / litmus)
Observation
- Formation of a coloured “lake” (red/blue coloured ppt)
This confirms Al³⁺
Iodide of Millon’s base is involved in the Nessler’s reagent test.
In the test for NH4+ (ammonium ion), iodide of Millon’s base is involved in the Nessler’s reagent test.
Nessler’s Reagent (used for detection):
- It contains alkaline solution of potassium tetraiodomercurate(II)
This is prepared using:
- Mercury salts
- Potassium iodide (KI)
- KOH
During preparation, iodide of Millon’s base type species are formed.
Reaction with :
When ammonium ion is present:
Observation:
- Brown precipitate forms
This precipitate is called:
Iodide of Millon’s base
Charcoal Cavity Test (CCT)
The charcoal cavity test is a dry test in qualitative inorganic analysis used to detect certain metal ions (cations) by heating them in a small cavity made in charcoal.
Procedure:
- A small cavity (hole) is made in a piece of charcoal
- A pinch of the salt is placed in it
- Heated strongly using a blowpipe flame
- Sometimes mixed with sodium carbonate (Na₂CO₃)
Principle:
- Charcoal acts as a reducing agent
- On heating, metal compounds are reduced to metal or metal oxide
- Different metals give characteristic residues / coatings
Observations (Important for exams):
| Metal ion | Observation |
|---|---|
| Zn²⁺ | Yellow when hot, white when cold (ZnO coating) |
| Pb²⁺ | Yellow coating (PbO) |
| Cu²⁺ | Red metallic bead (Cu) |
| Ag⁺ | Shiny white metallic bead |
| Bi³⁺ | Yellow coating |
Borax Bead Test — Full Concept for JEE / NEET
This is one of the most important dry tests in qualitative analysis to identify transition metal ions.
What is Borax?
Borax = Sodium tetraborate
Principle (CORE CONCEPT)
On heating, borax decomposes to form boric anhydride which reacts with metal oxides:
Step 1: Heating borax
= boric anhydride (glassy mass)
Step 2: Reaction with metal oxide
Forms colored metaborate bead
Why color appears?
- Due to transition metal ions
- Caused by d–d electronic transitions
Flame Types (VERY IMPORTANT)
| Flame type | Nature | Use |
|---|---|---|
| Oxidizing flame (O.F.) | Excess oxygen | Metal in higher oxidation state |
| Reducing flame (R.F.) | Less oxygen | Metal in lower oxidation state |
Same metal → different color in OF & RF
Important Colors (JEE/NEET GOLD)
| Metal ion | Oxidizing Flame | Reducing Flame |
|---|---|---|
| Cu²⁺ | Blue-green | Red (Cu₂O / Cu) |
| Co²⁺ | Deep blue | Deep blue |
| Cr³⁺ | Green | Green |
| Fe³⁺ | Yellow-brown | Bottle green |
| Mn²⁺ | Amethyst (purple) | Colorless |
| Ni²⁺ | Brown | Grey |
| V | Yellow | Green |
Procedure (Short)
- Take a platinum wire loop
- Dip in borax → heat → forms colorless bead
- Add salt → heat in O.F. & R.F.
- Observe color
Key Points (Exam Focus)
- Works mainly for transition metals
- Bead = transparent glassy mass
- Color depends on:
- Metal ion
- Oxidation state
- Flame type
- Cobalt always gives blue → very important MCQ
Common Mistakes Students Make
- Ignoring reducing flame colors
- Confusing Cu and Cr colors
- Forgetting Mn becomes colorless in R.F.
Different Zones of Flame (very important for JEE/NEET)
1. Inner Zone (Dark Zone)
- Location: Innermost part near burner
- Color: Dark / almost invisible
- Temperature: Lowest
- Nature: Unburnt gases present (no combustion)
Contains mixture of fuel gas + air
No practical use in tests
2. Middle Zone (Luminous Zone)
- Color: Yellow / bright
- Temperature: Moderate
- Nature: Reducing zone (incomplete combustion)
Contains glowing carbon particles
Gives sooty flame
Used in:
- Reduction reactions
- Sometimes in charcoal cavity test
3. Outer Zone (Non-luminous Zone)
- Color: Blue
- Temperature: Highest
- Nature: Oxidizing zone (complete combustion)
Contains excess oxygen
Clean flame (no soot)
Used in:
- Borax bead test (oxidizing flame)
- Flame test
- Most heating purposes
Brown Ring Test — for Nitrate
.
Reagents Used:
Fresh **Ferrous sulfate solution
Concentrated Sulfuric acid
Procedure:
Take the salt solution (suspected NO3−NO_3^-NO3−)
Add freshly prepared FeSO₄ solution
Carefully pour conc. H₂SO₄ along the side of the test tube
Two layers form
Observation:
A brown ring appears at the junction of the two layers
Confirms presence of nitrate ion
⚙️Principle (CORE CONCEPT):
Step 1: Reduction of nitrate
NO3−+3Fe2++4H+→NO+3Fe3++2H2ONO_3^- + 3Fe^{2+} + 4H^+ \rightarrow NO + 3Fe^{3+} + 2H_2ONO3−+3Fe2++4H+→NO+3Fe3++2H2O
Nitrate is reduced to nitric oxide (NO)
Step 2: Formation of brown complex
[Fe(H2O)5(NO)]2+[Fe(H_2O)_5(NO)]^{2+}[Fe(H2O)5(NO)]2+
is nitrosyl complex gives the brown color
Key Points (Exam Focus):
Test confirms NO3 – (not nitrite)
Fresh FeSO₄ is essential
Brown ring forms at junction, not throughout solution
Complex contains NO (nitric oxide) ligand
Common Mistakes:
Shaking the test tube → destroys layers
Using old FeSO₄ → no result
Confusing with nitrite rick:
“Nitrate → NO → Brown complex ring”
