1°, 2°, 3° Amines with HNO₂
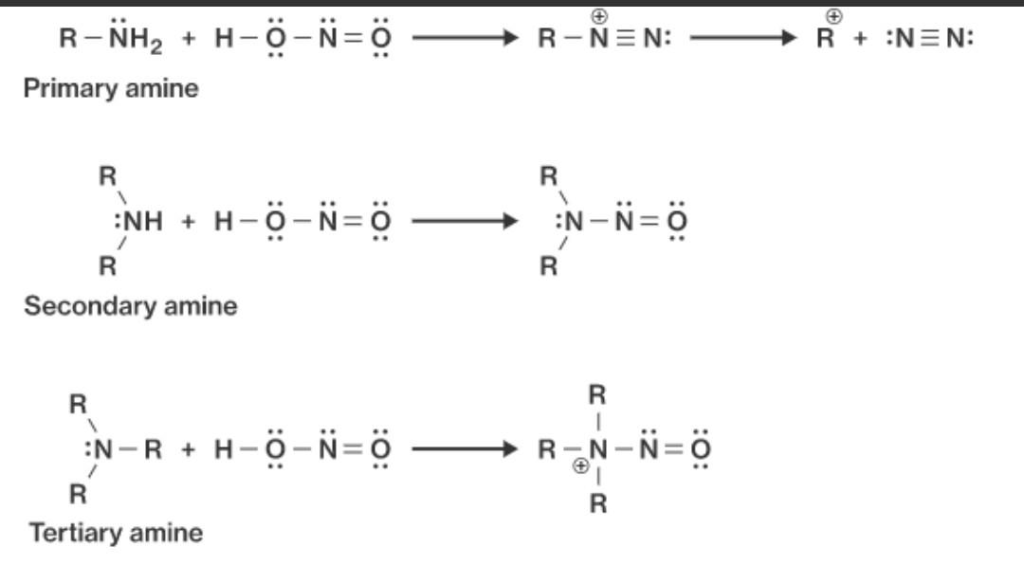
Primary (1°) Amine
(Just for comparison)
- Aliphatic:
Brisk effervescence (N₂ gas)
- Aromatic: forms diazonium salt
2° (Secondary) Amine
Reaction:
Product: N-nitrosamine
Observation:
- Yellow oily liquid
- No gas evolution
✔ Used as a test for 2° amines
3° (Tertiary) Amine
(A) Aliphatic 3° Amine:
Forms ammonium nitrite salt
Observation:
- No visible reaction
- No gas
(B) Aromatic 3° Amine:
Undergoes electrophilic substitution (ring reaction)
Example: aniline derivatives like dimethylaniline
✔ Green-colored solution (often)
Summary Table
| Amine Type | Product | Observation |
|---|---|---|
| 1° | Alcohol / Diazonium | N₂ gas |
| 2° | N-nitrosamine | Yellow oily liquid |
| 3° (aliphatic) | Salt | No reaction |
| 3° (aromatic) | p-nitroso compound | Colored solution |
Quick Trick
- 1° → Gas (N₂)
- 2° → Nitroso (yellow oil)
- 3° → No gas
Correct Reagent
We use NaNO₂ + HCl, but the actual reacting species is HNO₂ (nitrous acid)
Why?
- HNO₂ is unstable, so it is not stored directly
- It is generated in situ:
Conclusion
✔ In reaction/mechanism → write HNO₂
✔ In practical/lab conditions → write NaNO₂ + HCl
emperature is very important in reactions with nitrous acid (HNO₂ / NaNO₂ + HCl).
Required Temperature
✔ Primary (1°) Aromatic Amines
0–5°C (ice-cold conditions)
- Needed to form stable diazonium salt
- Above 5°C → diazonium salt decomposes
Primary (1°) Aliphatic Amines
Also done at 0–5°C
- But diazonium salt is unstable → immediately decomposes to alcohol + N₂
Secondary (2°) Amines
0–5°C preferred
- Forms N-nitrosamine
- Low temperature prevents side reactions
Tertiary (3°) Amines
- Aliphatic: 0–5°C (no major reaction, just salt formation)
- Aromatic: slightly higher temp possible, but usually cold conditions used
Summary Table
| Amine Type | Temperature |
|---|---|
| 1° Aromatic | 0–5°C (very important) |
| 1° Aliphatic | 0–5°C |
| 2° | 0–5°C |
| 3° | 0–5°C (generally) |
