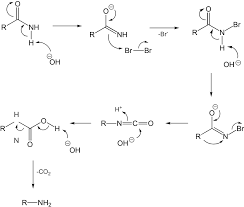
It is the conversion of an amide (R–CONH₂) into a primary amine (R–NH₂) with one carbon less using Br₂ + KOH/NaOH.
Overall Reaction
Key point: Chain length decreases by one carbon
Stepwise Mechanism
Step 1: Formation of N-bromoamide
Amide reacts with bromine in basic medium:
Step 2: Deprotonation
Step 3: Rearrangement (Key Step )
- Alkyl group (R) migrates from carbonyl carbon → nitrogen
- Br⁻ leaves
Forms isocyanate (R–N=C=O)
Step 4: Hydrolysis of Isocyanate
Step 5: Decarboxylation
Final Product
✔ Primary amine (R–NH₂)
✔ CO₂ released
Important Features
- ✔ One carbon less than starting amide
- ✔ Always gives 1° amine
- ✔ Rearrangement step is key (migration)
Quick Trick“CONH₂ → NH₂ (minus one carbon)”
Exam Points
- Migration order:
- Works only for primary amides
- Intermediate: Isocyanate (very important)