What is basic strength?
Amines act as bases because of the lone pair on nitrogen (N) that can accept a proton (H⁺)
Key Concepts Affecting Basic Strength
1. Availability of Lone Pair
- More available lone pair → stronger base
- If lone pair is involved in resonance or bonding → weaker base
2. Inductive Effect (+I and –I)
- Alkyl groups donate electrons (+I effect) → increase electron density on N → increase basicity
Order in gas phase:
But this changes in aqueous solution (see point 4).
3. Resonance Effect
- If lone pair participates in resonance → less available → weaker base
Example:
- Aniline is less basic because lone pair is delocalized in benzene ring
4. Solvation Effect (VERY IMPORTANT for NEET)
In aqueous solution, stability of protonated amine matters.
- More H-bonding → more stable → stronger base
- 1° amines are better solvated than 3° amines
Order in aqueous solution:
5. Steric Hindrance
- Bulky groups around N hinder protonation
- More crowding → weaker base
This is why 3° amines are weaker in water
6. Hybridization of Nitrogen
- More s-character → electrons closer to nucleus → less available → weaker base
7. Aromatic vs Aliphatic Amines
- Aliphatic amines → stronger base
- Aromatic amines (like aniline) → weaker due to resonance
Important Orders to Remember
Gas Phase:
Aqueous Solution:
Aromatic vs Aliphatic:
Basic strength in heterocyclic compounds
Core Idea (MOST IMPORTANT)
Is the nitrogen lone pair part of the aromatic system or not?
Case 1: Lone pair NOT involved in aromaticity → Basic
Example: Pyridine-type
- Nitrogen is sp² hybridized
- Lone pair lies in an sp² orbital (outside π-system)
- Available for protonation → basic
Example:
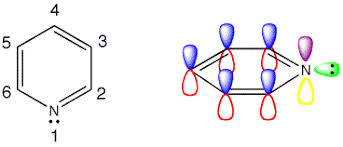
Pyridine
Case 2: Lone pair involved in aromaticity → Very weak base
Example: Pyrrole-type
- Lone pair is part of aromatic sextet (6π electrons)
- Not available for H⁺ → very weak base
Example:
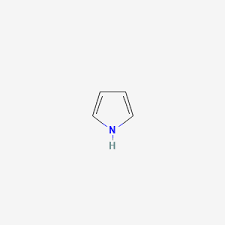
Pyrrole
Case 3: Multiple heteroatoms (competition effect)
Example: Imidazole, Pyrazole
- One N behaves like pyridine (basic)
- Other N behaves like pyrrole (non-basic)
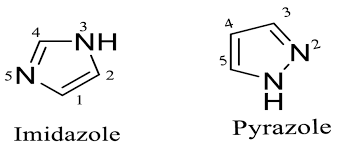
So overall: moderately basic
key Rule
The nitrogen whose lone pair is NOT involved in aromaticity is basic.
Imidazole
- N–H nitrogen (shown with H)
❌ Lone pair is part of aromatic sextet → NOT basic - Other nitrogen (without H)
✅ Lone pair is free → BASIC
Basic position = N without H (pyridine-like N)
Pyrazole
- N–H nitrogen
❌ Lone pair involved in aromaticity → NOT basic - Adjacent nitrogen (without H)
✅ Lone pair free → BASIC
Basic position = N without H
Important Examples & Order
1. Pyridine vs Pyrrole
✔ Pyridine → basic
✔ Pyrrole → almost non-basic
2. Aliphatic vs Aromatic Heterocycles
3. Imidazole (VERY IMPORTANT)
Reason: Extra stabilization of conjugate acid
Summary Table
| Compound Type | Lone Pair | Basic Strength |
|---|---|---|
| Pyridine | Free | Moderate |
| Pyrrole | In aromaticity | Very weak |
| Imidazole | One free | Moderate–strong |
Standard Basic Strength Order (Important)
Piperidine>Pyrrolidine>Imidazole>Pyridine>Aniline>Pyrazole>Pyrrole
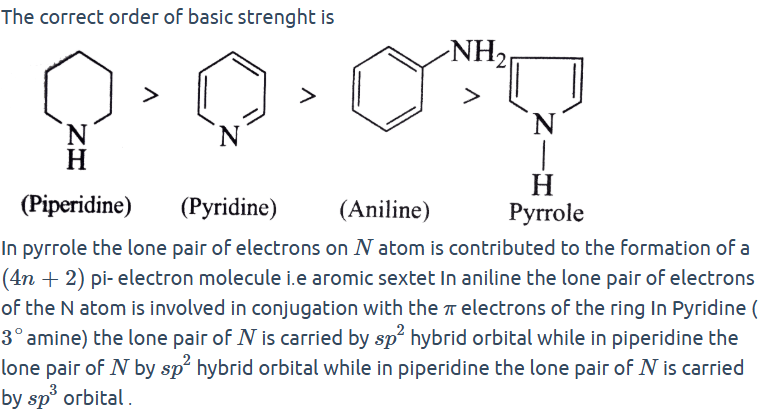
Answers:

