Hydrogenation of alkene is the addition of H₂ across a C=C double bond in presence of a metal catalyst like Ni, Pd, or Pt, forming an alkane.
1. Based on Substitution of Double Bond
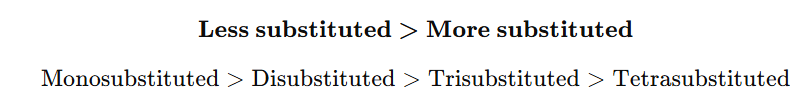
2.Cis vs Trans Alkenes
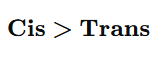
Reason:
- Cis isomer is less stable (higher energy)
- Therefore hydrogenates faster
3.Cyclic vs Acyclic
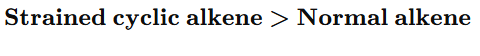
cyclopropane reacts faster due to ring strain.
4. Conjugated vs Isolated
Isolated double bond hydrogenates normally.
In conjugated systems, 1,4-addition may occur depending on conditions.
More stable alkene → Lower rate of hydrogenation why?
Hydrogenation is an exothermic reaction.
- More stable alkene = lower potential energy
- Therefore it releases less heat on hydrogenation
- Lower energy difference → smaller driving force
- Hence reacts more slowly
1. Which alkene hydrogenates fastest?
A) CH₃–CH=CH₂
B) CH₂=CH₂
C) (CH₃)₂C=CH₂
D) (CH₃)₂C=C(CH₃)₂
Answer: B
2. The rate of hydrogenation is inversely proportional to:
A) Heat of hydrogenation
B) Stability of alkene
C) Surface area of catalyst
D) Pressure of H₂
Answer: B
3. Which has lowest heat of hydrogenation?
A) 1-Butene
B) cis-2-Butene
C) trans-2-Butene
D) 2-Methyl-2-butene
Answer: D
4. Correct order of rate of hydrogenation:
A) Tetra > Tri > Di > Mono
B) Mono > Di > Tri > Tetra
C) Tri > Di > Mono > Tetra
D) Di > Mono > Tri > Tetra
Answer: B
5. Which reacts faster in hydrogenation?
A) trans-2-Butene
B) cis-2-Butene
C) Both equal
D) 2-Methyl-2-butene
Answer: B
6. Hydrogenation of alkene is:
A) Anti addition
B) Syn addition
C) Free radical reaction
D) Electrophilic addition
Answer: B
7. Which alkene is most stable?
A) CH₂=CH₂
B) CH₃–CH=CH₂
C) (CH₃)₂C=CH₂
D) (CH₃)₂C=C(CH₃)₂
Answer: D
8. Which will hydrogenate slowest?
A) 1-Butene
B) 2-Butene
C) 2-Methylpropene
D) 2,3-Dimethyl-2-butene
Answer: D
9. More stable alkene has:
A) Higher heat of hydrogenation
B) Lower heat of hydrogenation
C) Same heat of hydrogenation
D) Zero heat of hydrogenation
Answer: B
10. Strained cyclic alkene hydrogenates faster because:
A) It is more stable
B) It has lower energy
C) Relief of ring strain
D) It is more substituted
Answer: C
JEE/NEET Concept
Heat of hydrogenation order:
Higher heat release means less stable alkene → faster hydrogenation.
MCQs
1. Which cyclic alkene hydrogenates fastest?
A) Cyclohexene
B) Cyclopentene
C) Cyclobutene
D) Cyclopropene
Answer: D
2. The fastest hydrogenation is shown by the alkene having:
A) Maximum stability
B) Maximum ring strain
C) Maximum substitution
D) Minimum heat of hydrogenation
Answer: B
3. Arrange in increasing rate of hydrogenation:
A) Cyclohexene < Cyclopentene < Cyclobutene < Cyclopropene
B) Cyclopropene < Cyclobutene < Cyclopentene < Cyclohexene
C) Cyclobutene < Cyclopropene < Cyclohexene < Cyclopentene
D) Cyclopentene < Cyclohexene < Cyclobutene < Cyclopropene
Answer: A
4. Cyclohexene hydrogenates slower than cyclobutene because:
A) It is more substituted
B) It has less ring strain
C) It has higher energy
D) It forms unstable product
Answer: B
5. Heat of hydrogenation is maximum for:
A) Cyclohexene
B) Cyclopentene
C) Cyclobutene
D) Cyclopropene
Answer: D
Numerical-Type Concept Question
The heats of hydrogenation are:
- Cyclohexene → −119 kJ/mol
- Cyclopentene → −120 kJ/mol
- Cyclobutene → −127 kJ/mol
- Cyclopropene → −151 kJ/mol
Question:
Which is most stable? Which reacts fastest?
Solution:
✔ Most stable: Cyclohexene (least heat released)
✔ Fastest reaction: Cyclopropene (highest heat released)
“Hydrogenation rate of cyclic alkenes depends on ring strain. Smaller rings possess greater angle strain and hence higher heat of hydrogenation. Therefore, they react faster.”
JEE/NEET Important Order
Rate of hydrogenation:
Comparison Example (Very Important for Exams)
Compare:
- 1,3-Butadiene (conjugated)
- 1,4-Pentadiene (isolated)
Stability Order:
Rate of Hydrogenation:
JEE/NEET Important Order
Rate of hydrogenation:
(Benzene hydrogenates very slowly due to aromatic stability)
