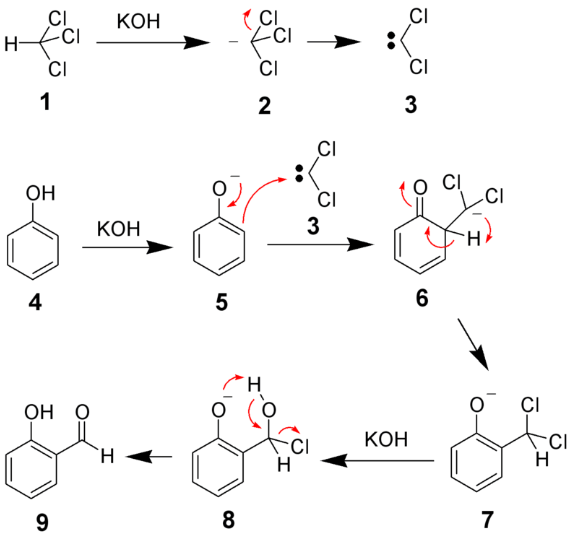
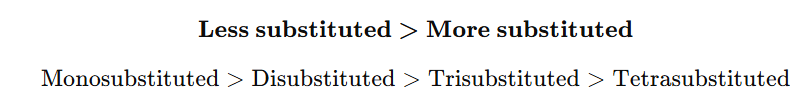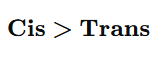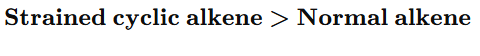Hydrogenation of alkene is the addition of H₂ across a C=C double bond in presence of a metal catalyst like Ni, Pd, or Pt , forming an alkane.
1. Based on Substitution of Double Bond
2.Cis vs Trans Alkenes
Reason:
Cis isomer is less stable (higher energy)
Therefore hydrogenates faster
3.Cyclic vs Acyclic
cyclopropane reacts faster due to ring strain.
4. Conjugated vs Isolated
Isolated double bond hydrogenates normally.
In conjugated systems, 1,4-addition may occur depending on conditions.
More stable alkene → Lower rate of hydrogenation why?
Hydrogenation is an exothermic reaction .
More stable alkene = lower potential energy
Therefore it releases less heat on hydrogenation
Lower energy difference → smaller driving force
Hence reacts more slowly
1. Which alkene hydrogenates fastest? A) CH₃–CH=CH₂
Answer: B
2. The rate of hydrogenation is inversely proportional to: A) Heat of hydrogenation
Answer: B
3. Which has lowest heat of hydrogenation? A) 1-Butene
Answer: D
4. Correct order of rate of hydrogenation: A) Tetra > Tri > Di > Mono
Answer: B
5. Which reacts faster in hydrogenation? A) trans-2-Butene
Answer: B
6. Hydrogenation of alkene is: A) Anti addition
Answer: B
7. Which alkene is most stable? A) CH₂=CH₂
Answer: D
8. Which will hydrogenate slowest? A) 1-Butene
Answer: D
9. More stable alkene has: A) Higher heat of hydrogenation
Answer: B
10. Strained cyclic alkene hydrogenates faster because: A) It is more stable
Answer: C
JEE/NEET Concept
Heat of hydrogenation order:Cyclopropene > Cyclobutene > Cyclopentene > Cyclohexene \text{Cyclopropene > Cyclobutene > Cyclopentene > Cyclohexene}
Higher heat release means less stable alkene → faster hydrogenation .
MCQs
1. Which cyclic alkene hydrogenates fastest? A) Cyclohexene
Answer: D
2. The fastest hydrogenation is shown by the alkene having: A) Maximum stability
Answer: B
3. Arrange in increasing rate of hydrogenation: A) Cyclohexene < Cyclopentene < Cyclobutene < Cyclopropene
Answer: A
4. Cyclohexene hydrogenates slower than cyclobutene because: A) It is more substituted
Answer: B
5. Heat of hydrogenation is maximum for: A) Cyclohexene
Answer: D
Numerical-Type Concept Question
The heats of hydrogenation are:
Cyclohexene → −119 kJ/mol
Cyclopentene → −120 kJ/mol
Cyclobutene → −127 kJ/mol
Cyclopropene → −151 kJ/mol
Question:
Which is most stable? Which reacts fastest?
Solution:
✔ Most stable: Cyclohexene (least heat released)Fastest reaction: Cyclopropene (highest heat released)
“Hydrogenation rate of cyclic alkenes depends on ring strain. Smaller rings possess greater angle strain and hence higher heat of hydrogenation. Therefore, they react faster.”
JEE/NEET Important Order
Rate of hydrogenation:Isolated diene > Conjugated diene > Aromatic compound \textbf{Isolated diene > Conjugated diene > Aromatic compound}
Comparison Example (Very Important for Exams)
Compare:
1,3-Butadiene (conjugated)
1,4-Pentadiene (isolated)
Stability Order:
Conjugated > Isolated \textbf{Conjugated > Isolated}
Rate of Hydrogenation:
Isolated > Conjugated \textbf{Isolated > Conjugated}
JEE/NEET Important Order
Rate of hydrogenation:Isolated diene > Conjugated diene > Aromatic compound \textbf{Isolated diene > Conjugated diene > Aromatic compound}
(Benzene hydrogenates very slowly due to aromatic stability)