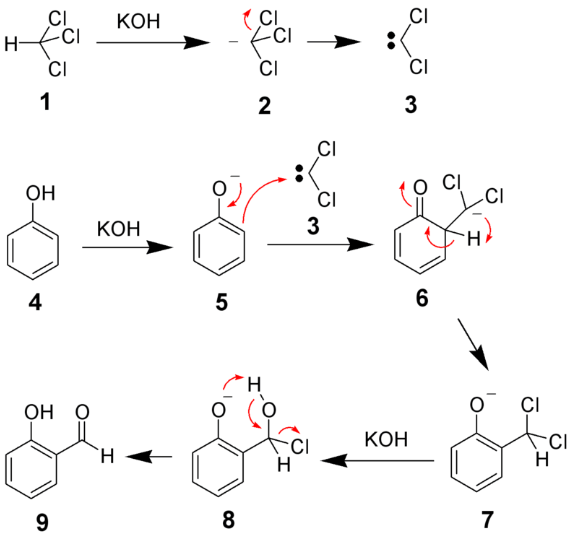
The Reimer–Tiemann reaction is used to introduce a –CHO (aldehyde) group into a phenol ring, usually at the ortho position (forming salicylaldehyde).
Overall reaction
Phenol + CHCl₃ + NaOH → o-hydroxybenzaldehyde (major) + p-hydroxybenzaldehyde (minor)
Key Points to Remember
- Reagent: CHCl₃ + NaOH
- Reactive intermediate: dichlorocarbene (:CCl₂)
- Directing group: –O⁻ (phenoxide) → ortho/para directing
- Major product: ortho-hydroxybenzaldehyde
- Para product is minor due to intramolecular H-bonding stabilizing ortho product
