Hydrocarbons form the fundamental backbone of Organic Chemistry, consisting only of carbon and hydrogen atoms.
They are broadly classified into alkanes, alkenes, alkynes, and aromatic hydrocarbons based on bonding and structure.
Understanding their properties and reactions is crucial for mastering concepts in competitive exams like JEE and NEET.
This test paper is designed to evaluate conceptual clarity, problem-solving ability, and application skills.
It covers important topics such as nomenclature, isomerism, and reaction mechanisms of hydrocarbons.
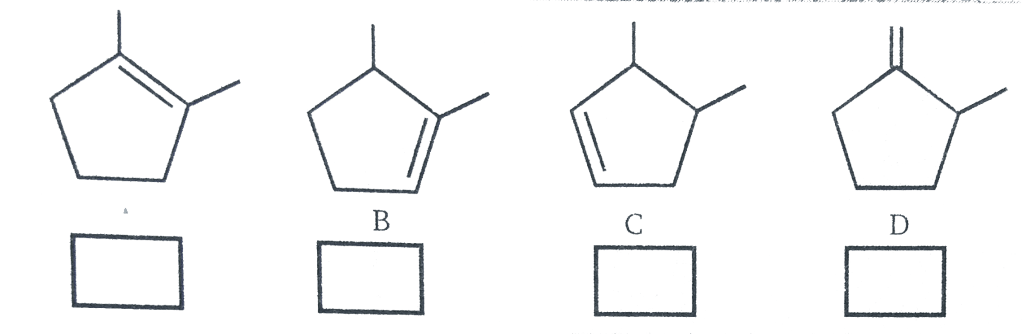
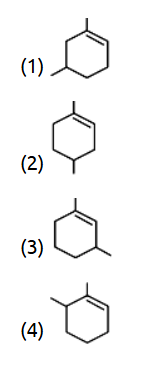
- Arrange the following alkenes in order of their stability
2. But-2-yne and hydrogen (one mole each) are separately treated with (i) P d / C and (ii) N a / liq. N H 3 to give the products X and Y respectively
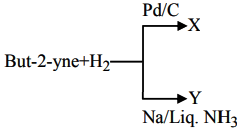
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of X is zero
C. Boiling point of X is higher than Y .
D. X and Y react with O 3 / Z n + H 2 O to give different products.
Choose the correct answer from the options given below :
(1) B and C only
(2) B and D only
(3) A and B only
(4) A and C only
3.
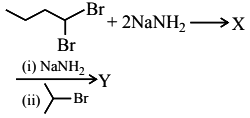
The product Y formed is :
(1) 2-methylhex-2-yne
(2) 5-methylhex-2-yne
(3) 2-methylhex-3-yne
(4) Isopropylbut-1-yne
4
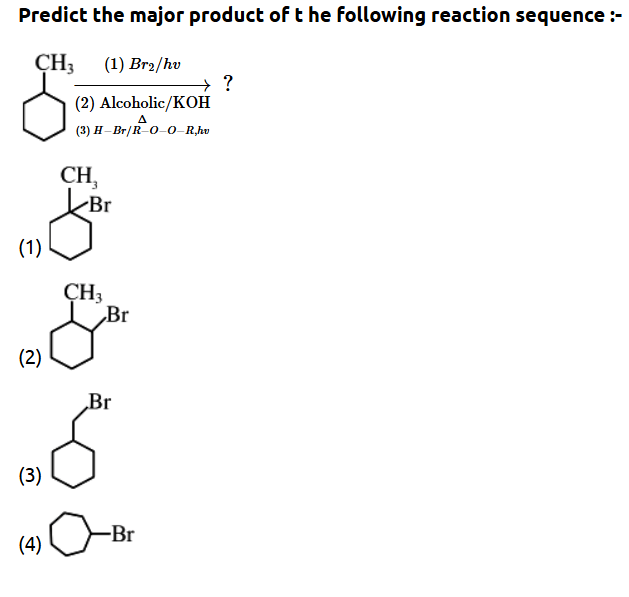
5. Which compound would give 3-methyl-6- oxoheptanal upon ozonolysis ?
6. Given below are two statements :
Statement (I) : Neopentane forms only one monosubstituted derivative.
Statement (II) : Melting point of neopentane is higher than n-pentane.
In the light of the above statements, choose the most appropriate answer from the options given below :
(1) Statement I is correct but Statement II is incorrect
(2) Both Statement I and Statement II are correct
(3) Both Statement I and Statement II are incorrect
7. Total number of sigma (σ) and pi(π) bonds respectively present in hex-1-en-4-yne are:
(1) 13, 3
(2) 14, 3
(3) 3, 14
(4) 14, 13
8. Identify product [A], [B] and [C] in the following reaction sequence.
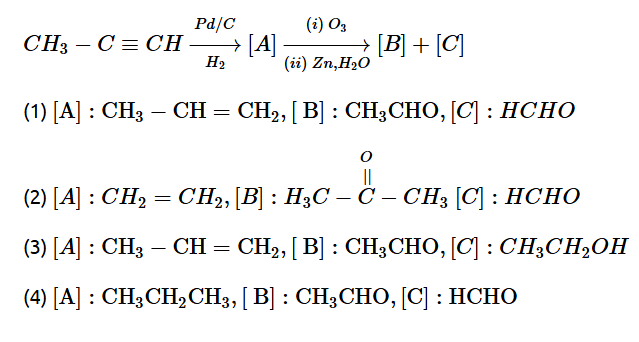
9.Statement I : Nitration of benzene involves the following step –
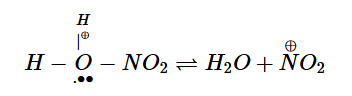
Statement II: Use of Lewis base promotes the electrophilic substitution of benzene. In the light of the above statements, choose the most appropriate answer from the options given below :
(1) Both Statement I and Statement II are incorrect
(2) Statement I is correct but Statement II is incorrect (3) Both Statement I and Statement II are correct (4) Statement I is incorrect but Statement II is correct
10.Compound A formed in the following reaction reacts with B gives the product C. Find out A and B
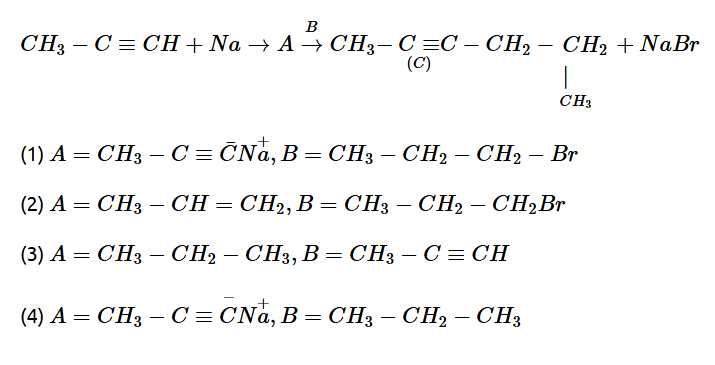
11. Product ‘A’ of following sequence of reactions is
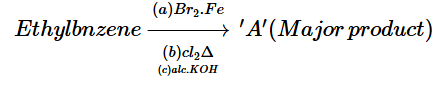
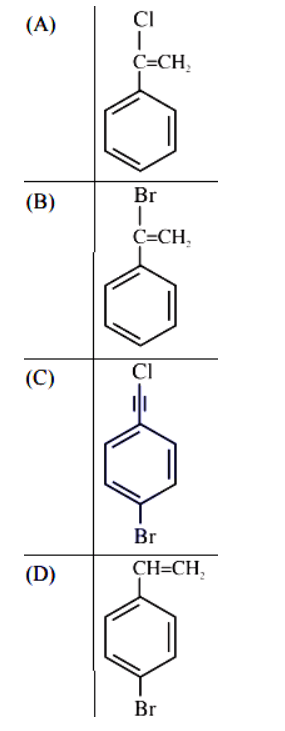
12
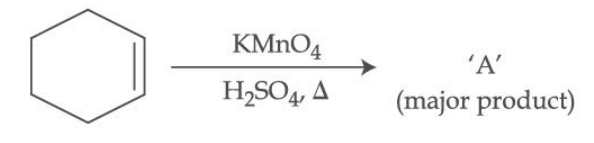
A) Cyclohexanol
B) Hexane-1,6-dioic acid (Adipic acid)
C) Cyclohexanone
D) 1,2-diol (Glycol)
13.Consider the following reaction :
Which of these reaction(s) will not produce Saytzeff product ?
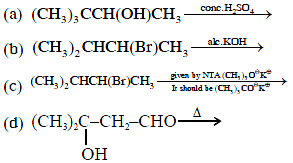
Which of these reaction(s) will not produce Saytzeff product ? (
1) (c) only
(2) (a) (c) and (d)
(3) (d) only
(4) (b) and (d)
15. In the following skew conformation of ethane, H’–C–C–H” dihedral angle is :
In the following skew conformation of ethane, H’–C–C–H” dihedral angle is :
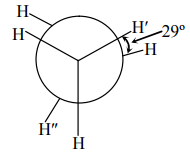
(1) 120º
(2) 58º
(3) 151º
(4) 149º
