Stereoisomerism organic chemistry JEE/NEET Concepts
What is Stereoisomerism?
Compounds with:
- Same molecular formula
- Same connectivity
- Different 3D arrangement
Types of Stereoisomerism
(A) Geometrical Isomerism
(B) Optical Isomerism
Geometrical Isomerism (cis–trans / E–Z)
Condition:
- Restricted rotation (C=C, ring)
- Each carbon must have two different groups
Types:
cis–trans (simple case)
- Same groups → cis
- Opposite → trans
Example:
- cis-2-butene vs trans-2-butene
E–Z system (priority based)
Use Cahn–Ingold–Prelog priority rules
- Higher priority same side → Z
- Opposite side → E
Priority Rules
Based on Cahn–Ingold–Prelog priority rules
When to use E–Z?
Use E–Z when:
- Alkene has 4 different groups
- cis–trans is not applicable
Priority Rules (STEP-BY-STEP)
Rule 1: Atomic Number
Higher atomic number = higher priority
Example:
- I > Br > Cl > F
- O > N > C > H
Rule 2: If same atom → go to next atom
Compare atoms attached to that atom
Example:
- –CH₃ vs –CH₂CH₃
\ Compare next atoms → ethyl gets higher priority
Rule 3: Multiple Bonds
Treat as if atom is bonded to duplicate atoms
Example:
- C=O → C attached to O, O
- C≡N → C attached to N, N, N
Rule 4: Isotopes
Higher mass number = higher priority
Example:
- D > H
MCQs (E–Z Configuration – Advanced)
Q1.
The correct configuration of the alkene is:
CH₃–CH = C(Cl)–CH₂CH₃
(A) E
(B) Z
(C) Cannot be determined
(D) No geometrical isomerism
Q2.
Which of the following has Z-configuration?
(A) CH₃–CH=CH–Br
(B) CH₃–CH=CH–CH₃
(C) CH₃–CH=CH–Cl
(D) CH₃–CH=CH–F
Q3.
The compound having E-configuration is:
(A) CH₃–CH=CH–NO₂
(B) CH₃–CH=CH–CN
(C) CH₃–CH=CH–CHO
(D) CH₃–CH=CH–CH₃
Q4.
Consider:
CH₃–CH = C(CH₃)–CHO
Its configuration is:
(A) E
(B) Z
(C) Both possible
(D) No GI
Q5.
Which has highest priority group correctly identified?
(A) –OH > –NH₂
(B) –CH₃ > –Cl
(C) –Br > –I
(D) –H > –D
Q6.
In the compound:
CH₃–C(Br)=C(Cl)–CH₂OH
The configuration is:
(A) E
(B) Z
(C) No GI
(D) Cannot decide
Q7.
Which shows no geometrical isomerism?
(A) CH₃–CH=CH–CH₃
(B) CH₂=CH–CH₃
(C) CHCl=CHCl
(D) CH₃–CH=CH–Cl
Q8.
In:
CH₃–CH = C(CN)–COOH
Configuration is:
(A) E
(B) Z
(C) Both
(D) None
Q9.
Priority order is correct for:
(A) –COOH > –CHO
(B) –CN > –COOH
(C) –CH₂OH > –CH₃
(D) –NH₂ > –OH
Q10.
In:
CH₃–CH = C(Br)–C≡N
Configuration is:
(A) E
(B) Z
(C) No GI
(D) Cannot be determined
Here are the correct options only
- A
- C
- A
- A
- A
- A
- B
- B
- C
- A
Optical Isomerism
Based on Chirality
A molecule is chiral if:
- It is non-superimposable mirror image
Chiral Carbon (asymmetric carbon)
- Attached to 4 different groups
Types:
(1) Enantiomers
- Mirror images
- Rotate plane polarized light in opposite directions
(2) Diastereomers
- Not mirror images
- Different physical properties
Meso Compounds
- Have chiral centers but optically inactive
- Due to internal plane of symmetry
Optical Activity
d(+) → dextrorotatory
l(–) → levorotatory
Optical Activity of Diastereomers
Case 1: Optically Active
If molecule:
- Has chiral center(s)
- No plane of symmetry
✔ Then diastereomer rotates plane polarized light
Case 2: Optically Inactive
If molecule:
- Has internal plane of symmetry (meso form)
✔ Then diastereomer is optically inactive
Example
Tartaric acid:
- (R,R) and (S,S) → Enantiomers → optically active
- (R,S) → Meso form → optically inactive
here:
- (R,R) vs (R,S) = Diastereomers
- But:
- (R,R) → active
- (R,S) → inactive
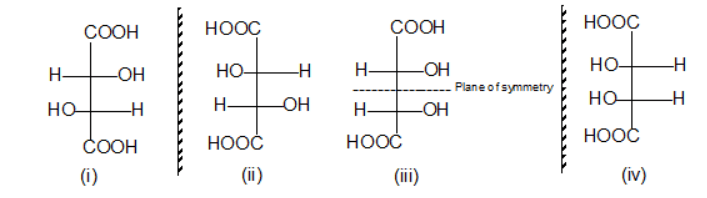
Final Answer
Enantiomers:
(i) & (ii)
Meso:
(iii)
Diastereomers:
- (i) & (iii)
- (ii) & (iii)
- (i) & (iv)
- (ii) & (iv)
- (iii) & (iv)
Key Recall
- (i) & (ii) → enantiomers → both optically active
- (iii) → meso → optically inactive
- (iv) → optically active
Diastereomer Pairs Analysis
(i) & (iii)
- (i) → active
- (iii) → inactive (meso)
Pair: one active + one inactive
(ii) & (iii)
- (ii) → active
- (iii) → inactive
Pair: one active + one inactive
(iii) & (iv)
- (iii) → inactive
- (iv) → active
Pair: one active + one inactive
(i) & (iv)
- (i) → active
- (iv) → active
Pair: both optically active
(ii) & (iv)
- (ii) → active
- (iv) → active
Pair: both optically active
3. Effect on number of stereoisomers
General:
With plane of symmetry:
Some structures become identical (meso)
How to find the plane of symmetry in biphenyls and substituted cyclohexane?
Optical activity is shown by compounds in which plane of symmetry is not present, if plane of symmetry is present in any compound then they will never show optical activity.
Optical activity is a kind of property of any compound which rotates the plane polarized light to the right or left side from the direction of upcoming light.
In biphenyls two benzene rings are present and if a plane of symmetry is present within the molecule then they don’t show optical activity.
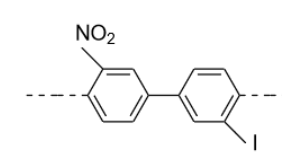
This molecule does not have a plane of symmetry
Biphenyl rotation:
- Structure: Biphenyl has two benzene rings connected by a single C–C bond.
- So, in principle, it’s a single bond, which allows rotation.
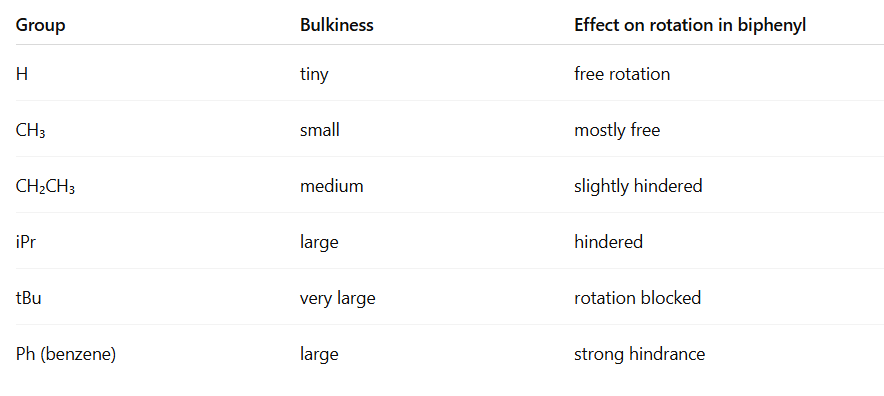
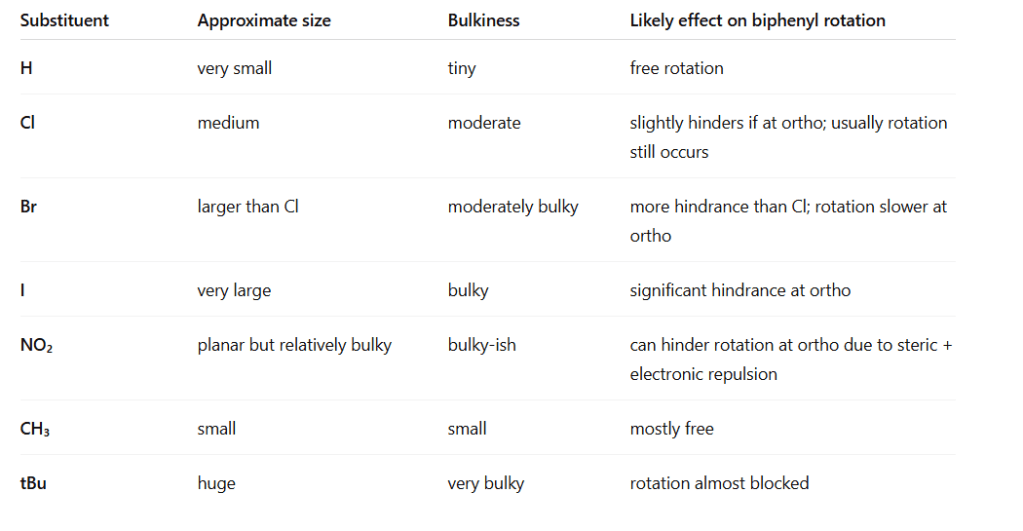
- Rotation limitation:
- Each benzene ring is planar.
- If there are no substituents at ortho positions, the two rings can rotate freely at room temperature.
- If there are bulky groups at the ortho positions (next to the connecting bond), steric hindrance prevents free rotation, and the molecule may adopt a twisted conformation.
- Extremely hindered biphenyls can even be atropisomers (stable enantiomers due to restricted rotation)
. It is chiral if the biphenyl can’t rotate freely (restricted rotation around the central C–C bond) and may exist as atropisomers.
Position matters most
- Only ortho substituents (2,2’ positions on biphenyl) cause steric hindrance.
- Meta and para positions do not block rotation.
