Step 1: Calculate DU
Formula:
For C₆H₁₄O (ignore O in DU):
Meaning of DU = 0
- No double bond
- No triple bond
- No ring
So, all structures are open-chain saturated alcohols

Formula:
For C₆H₁₄O (ignore O in DU):
So, all structures are open-chain saturated alcohols
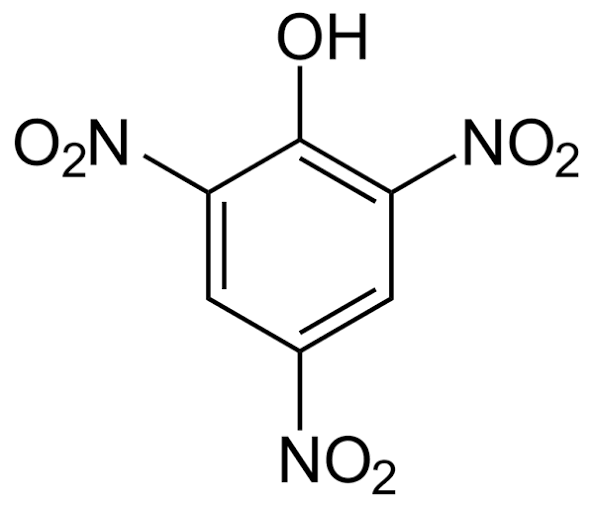
HCOOH (Picric Acid)
Picric acid (2,4,6-trinitrophenol) is a strong acid due to the presence of three –NO₂ groups, which strongly withdraw electrons and stabilize the phenoxide ion. Because of this high acidity, it can react even with a weak base like NaHCO₃.
Reaction:
Picric acid + NaHCO₃ → Sodium picrate (soluble) + CO₂ + H₂O
So, it dissolves in NaHCO₃ with effervescence of CO₂ gas.
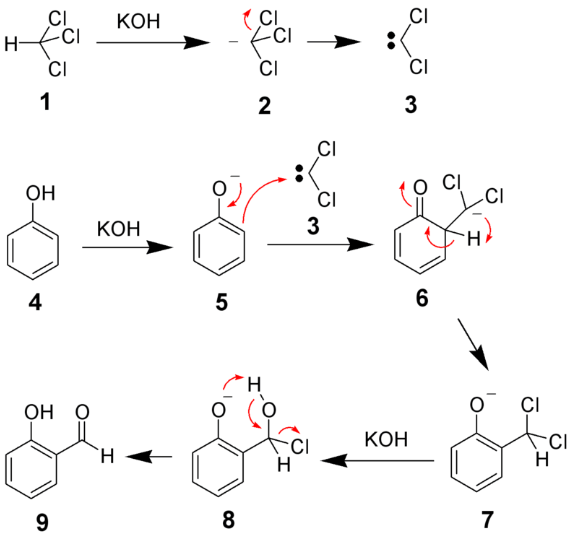
The Reimer–Tiemann reaction is used to introduce a –CHO (aldehyde) group into a phenol ring, usually at the ortho position (forming salicylaldehyde).
Phenol + CHCl₃ + NaOH → o-hydroxybenzaldehyde (major) + p-hydroxybenzaldehyde (minor)
According to Bohr, electrons move in stationary orbits where they do not radiate energy, hence energy of an orbit remains constant with time.
Bohr assumed stationary orbits (not proved mathematically) This explains atomic stability Energy is quantized and constant in each orbit.
Electrostatic force = Centripetal force
Step 1: Electron is attracted to nucleus, so:
Step 2 : Quantization of angular momentum
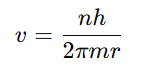
Step 3: Substitute v into Step 1
Step 4: Simplify
Step 5: Solve for radius r
Simplify:
Final Result
where,
Key Results
Bohr model, hydrogen-like atom, radius derivation, angular momentum quantization, electrostatic force
The line corresponding to the transition where the electron comes from an infinitely large orbit (n = ∞) to a fixed lower energy level.
For limiting line
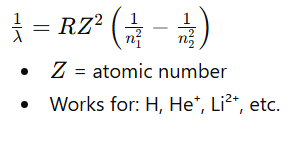
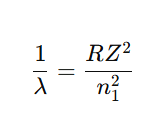
Limiting line = shortest wavelength (maximum energy) line of a series.
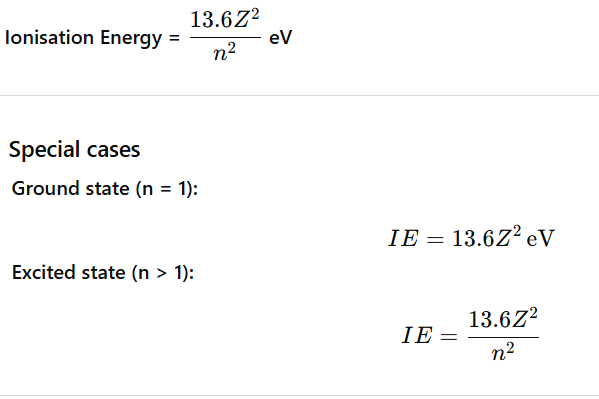
The Lyman α (alpha) line is the first spectral line of the Lyman series.
Definition: Oxides that react with water to form acids or react with bases to give salt + water.
Examples:
Reactions:
Trend:
Definition: Oxides that react with water to form bases or react with acids to give salt + water.
Examples:
Reactions:
Definition: Oxides that react with both acids and bases.
Examples (VERY IMPORTANT for exams):
4. Neutral Oxides
Definition: Oxides that do not react with acids or bases.
Examples:
IMPORTANT PERIODIC TREND (MOST ASKED)
Basic → Amphoteric → Acidic
Example (Period 3):
Na₂O → MgO → Al₂O₃ → SiO₂ → P₂O₅ → SO₃ → Cl₂O₇
Key shift:
| Type | Nature | Examples |
|---|---|---|
| Basic | Metal oxides | Na₂O, CaO |
| Amphoteric | Both | Al₂O₃, ZnO |
| Acidic | Non-metal oxides | SO₃, CO₂ |
| Neutral | No reaction | CO, NO |
EXAM TIPS
Q1. Arrange in increasing acidic nature:
A. CO₂ < SiO₂ < SO₃
B. SiO₂ < CO₂ < SO₃
C. SO₃ < CO₂ < SiO₂
D. CO₂ < SO₃ < SiO₂
Q2. Arrange in decreasing basic strength:
A. Na₂O > MgO > Al₂O₃
B. Al₂O₃ > MgO > Na₂O
C. MgO > Na₂O > Al₂O₃
D. Na₂O > Al₂O₃ > MgO
Q3. Arrange in increasing amphoteric character:
A. Na₂O < MgO < Al₂O₃
B. Al₂O₃ < MgO < Na₂O
C. MgO < Na₂O < Al₂O₃
D. Na₂O < Al₂O₃ < MgO
Q4. Arrange in increasing acidic strength:
A. SO₂ < SO₃ < Cl₂O₇
B. Cl₂O₇ < SO₃ < SO₂
C. SO₃ < SO₂ < Cl₂O₇
D. SO₂ < Cl₂O₇ < SO₃
Q5. Arrange in increasing basic character:
A. CaO < MgO < Na₂O
B. Na₂O < MgO < CaO
C. MgO < CaO < Na₂O
D. CaO < Na₂O < MgO
Q6. Arrange in increasing acidic nature:
A. P₂O₅ < SO₃ < Cl₂O₇
B. Cl₂O₇ < SO₃ < P₂O₅
C. SO₃ < P₂O₅ < Cl₂O₇
D. P₂O₅ < Cl₂O₇ < SO₃
Q7. Arrange in increasing basic strength:
A. BeO < MgO < CaO
B. CaO < MgO < BeO
C. MgO < BeO < CaO
D. BeO < CaO < MgO
Q8. Arrange in increasing acidic character:
A. N₂O₃ < NO₂ < N₂O₅
B. N₂O₅ < NO₂ < N₂O₃
C. NO₂ < N₂O₃ < N₂O₅
D. N₂O₃ < N₂O₅ < NO₂
ANSWERS
SHORT TRICKS (VERY IMPORTANT)
Electronegativity is the tendency of an atom to attract the shared pair of electrons towards itself in a chemical bond.
Important Points
Across a Period (→)
Increases
Down a Group (↓)
Decreases
🔹 Important Order
Fluorine (F) is the most electronegative element
More s-character → electrons closer to nucleus
The Pauling scale is the most commonly used scale to measure electronegativity, proposed by Linus Pauling.
Basic Idea
Electronegativity is calculated based on:
✔ Bond energies (bond dissociation enthalpies)
Formula (Conceptual)
Pauling related electronegativity difference to bond energy:
Where:
Key Points
| Element | Electronegativity |
|---|---|
| F | 4.0 |
| O | 3.5 |
| N | 3.0 |
| Cl | 3.0 |
| H | 2.1 |
Important Applications
1. Predict Bond Type
2. Predict Polarity
The Mulliken scale was proposed by Robert S. Mulliken.
Basic Idea
Electronegativity depends on:
✔ Ionisation Enthalpy (IE)
✔ Electron Gain Enthalpy (EGE / Electron Affinity)
So, it considers both:
🔹 Formula
(In some books, electron affinity is used instead of EGE)
🔹 Meaning
✔ So, higher value = higher electronegativity
🔹 Key Points
| Feature | Mulliken | Pauling |
|---|---|---|
| Based on | IE + EGE | Bond energy |
| Type | Absolute | Relative |
| Concept | Atomic property | Bond property |
The two scales are related approximately by:
Electron Gain Enthalpy is the enthalpy change when an electron is added to an isolated gaseous atom.
🔹 General Trends
EGE becomes more negative
Reason:
Down a Group (Top → Bottom)
EGE becomes less negative
Reason:
EGE ≈ zero or slightly positive
Reason:
❗ Exception 2: N (Group 15)
EGE is less negative than expected
Reason:
❗ Exception 3: Noble Gases (Group 18)
EGE is positive
Reason:
❗ Exception 4: Fluorine vs Chlorine
👉 Cl has more negative EGE than F ❗
❌ Expected: F > Cl
✅ Actual: Cl > F
Reason:
🔹 Important Order Example
Among halogens:
Cl > F > Br > I
👉 Adding 2nd electron:
❗ Always positive
Reason:
Increases down the group
O < S < Se < Te
Why is the 2nd Electron Gain Enthalpy of Oxygen Positive?
Consider the process:
🔹 Key Reason: Electron–Electron Repulsion
After gaining one electron:
Now, adding another electron:
✔ So, energy must be supplied → EGE becomes positive
Isoelectronic species are:
Atoms, ions, or molecules that have the same number of electrons.
🔹 Examples
All of these have 10 electrons → so they are isoelectronic
How to identify?
Just count total electrons
Example:
✔ Same electrons → Isoelectronic
🔹 Important Trend (VERY IMPORTANT for JEE/NEET)
In an isoelectronic series:
Size decreases as nuclear charge (Z) increases
For species with 10 electrons:
N³⁻ > O²⁻ > F⁻ > Ne > Na⁺ > Mg²⁺
Reason:
🔹 Key Exam Line
“Isoelectronic species have same number of electrons but different nuclear charge.”
The Van der Waals radius is defined as:
Half of the distance between the nuclei of two non-bonded identical atoms when they are just touching each other.
Example
If two neon atoms (not bonded) are 3.2 Å apart:
Van der Waals radius > Metallic radius > Covalent radius
| Radius Type | Condition | Smaller/Larger |
|---|---|---|
| Covalent radius | Bonded atoms | Smaller |
| Metallic radius | Metal lattice | Medium |
| Van der Waals | Non-bonded atoms | Largest |