HO–CH₂–CH₂–OH + HIO₄ → 2 HCHO (formaldehyde) + HIO₃ + H₂O
Explanation (2–3 marks)
- Periodic acid cleaves –CHOH–CHOH (vicinal diol)
- C–C bond breaks
- Each carbon forms a carbonyl compound
- Since both carbons are –CH₂OH → gives 2 molecules of formaldehyde

HO–CH₂–CH₂–OH + HIO₄ → 2 HCHO (formaldehyde) + HIO₃ + H₂O
Explanation (2–3 marks)
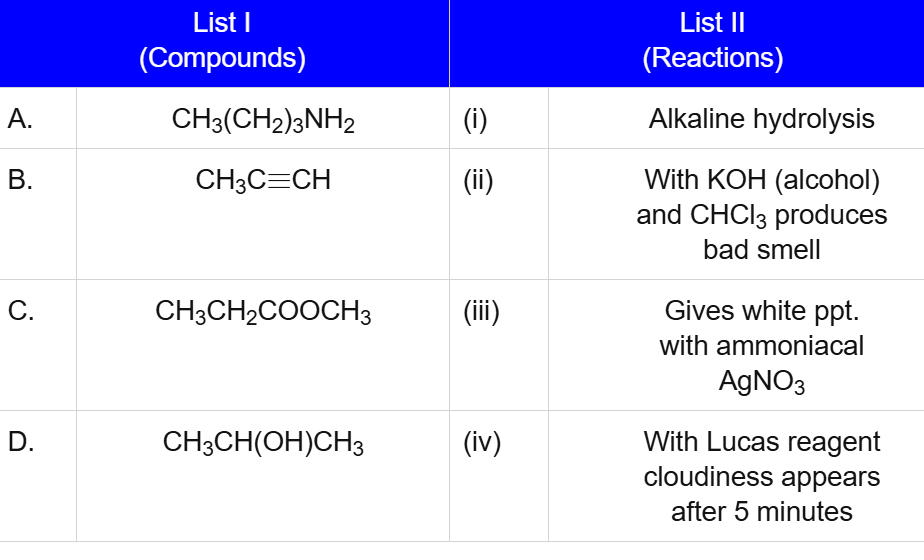
A. CH₃(CH₂)₃NH₂ → (i) Alkaline hydrolysis
B. CH₃C≡CH → (ii) With KOH (alcohol) and CHCl₃ produces bad smell
C. CH₃CH₂COOCH₃ → (iii) Gives white ppt. with ammoniacal AgNO₃
D. CH₃CH(OH)CH₃ → (iv) With Lucas reagent cloudiness
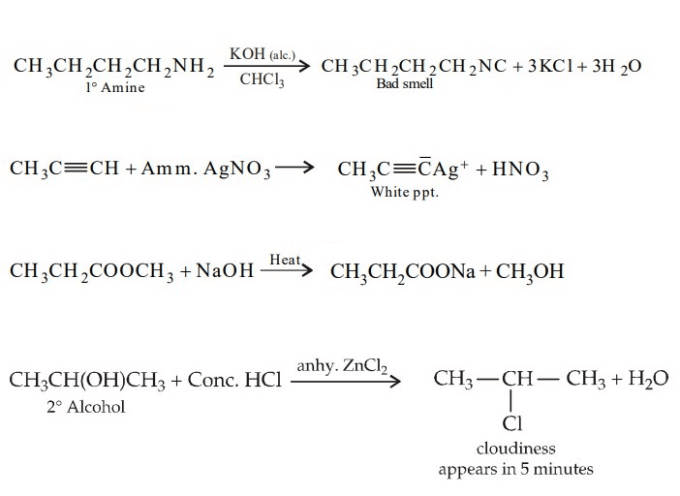
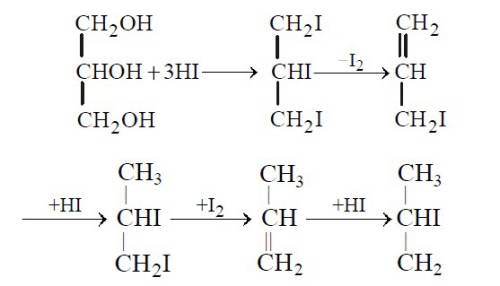
Glycerol + excess HI → 2-iodopropane (isopropyl iodide) + I₂ + H₂O
Explanation
Ethylene glycol (ethane-1,2-diol)
✔ Used as antifreeze because it prevents water from freezing in cold conditions.
Other than ethylene glycol, there are a few compounds used as antifreeze:
Other Antifreeze Compounds
1. Propylene glycol (propane-1,2-diol)
2. Glycerol (glycerine, propane-1,2,3-triol)
Formula:
For C₆H₁₄O (ignore O in DU):
So, all structures are open-chain saturated alcohols
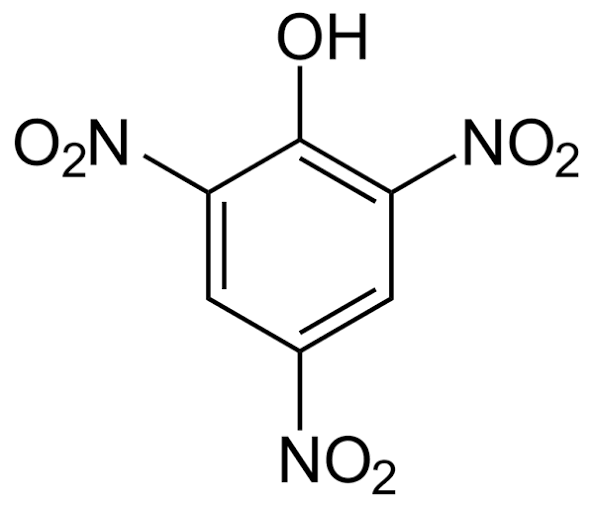
HCOOH (Picric Acid)
Picric acid (2,4,6-trinitrophenol) is a strong acid due to the presence of three –NO₂ groups, which strongly withdraw electrons and stabilize the phenoxide ion. Because of this high acidity, it can react even with a weak base like NaHCO₃.
Reaction:
Picric acid + NaHCO₃ → Sodium picrate (soluble) + CO₂ + H₂O
So, it dissolves in NaHCO₃ with effervescence of CO₂ gas.
CH₃CN > CH₃OH
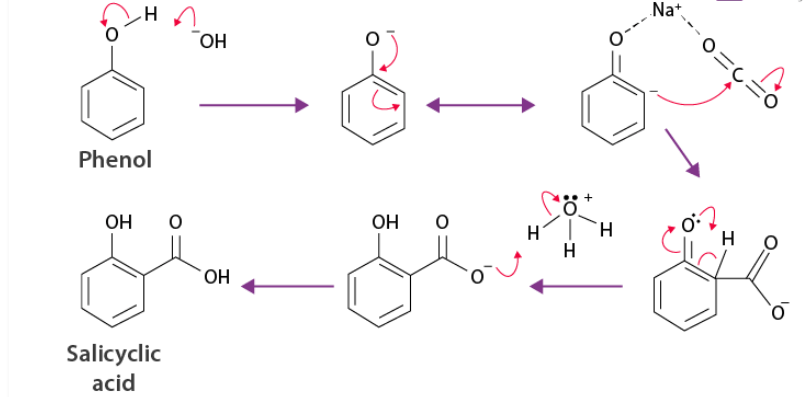
The Kolbe’s reaction (Kolbe–Schmitt reaction) is used to introduce a –COOH group into phenol, mainly at the ortho position.
Overall reaction
Phenol + NaOH + CO₂ (pressure, heat) → o-hydroxybenzoic acid (salicylic acid)
Key Points
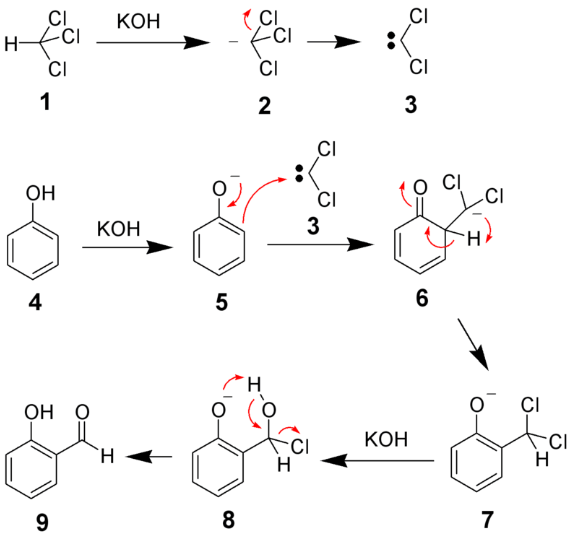
The Reimer–Tiemann reaction is used to introduce a –CHO (aldehyde) group into a phenol ring, usually at the ortho position (forming salicylaldehyde).
Phenol + CHCl₃ + NaOH → o-hydroxybenzaldehyde (major) + p-hydroxybenzaldehyde (minor)
Alkene → Alcohol (Markovnikov addition, no rearrangement)
CH₃–CH=CH₂ → CH₃–CHOH–CH₃
(Propene → 2-propanol)
Key Features (VERY IMPORTANT)
1. Markovnikov Addition
2. NO Carbocation Rearrangement ❌
This is the most asked point in JEE
Unlike acid-catalyzed hydration:
Alkene + Hg(OAc)₂ → cyclic intermediate
H₂O attacks more substituted carbon
NaBH₄ replaces Hg with H
Important Comparison (JEE trap)
| Reaction | Rearrangement | Markovnikov | Intermediate |
|---|---|---|---|
| Oxymercuration | ❌ No | ✔ Yes | Mercurinium ion |
| Acid hydration | ✔ Yes | ✔ Yes | Carbocation |
Special Case (VERY IMPORTANT)
Hg(OAc)₂ / ROH → Ether formation
👉 Alkene → Ether (Markovnikov)
Tough Concept Question
Because intermediate is mercurinium ion (bridged), not free carbocation
Memory Trick:
Mercury locks structure”
→ No rearrangement possible
Exam Trap
3° carbocation possible after rearrangement