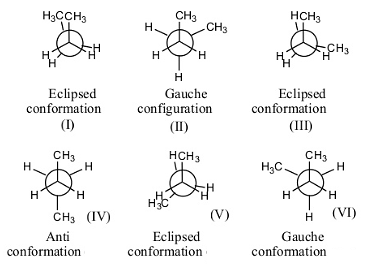
Types of Conformations of Butane
1. Anti Conformation (Most Stable)
- Dihedral angle = 180°
- Two CH₃ groups are opposite
- Minimum steric hindrance
Stability: Highest
✔ No torsional strain
✔ No steric crowding
2. Gauche Conformation
- Dihedral angle = 60°
- CH₃ groups are close but not eclipsed
Stability: Moderate
✔ Some steric hindrance
✔ Still relatively stable
3. Eclipsed Conformations (Unstable)
(a) CH₃–H Eclipsed
- Dihedral angle = 120° or 240°
- CH₃ eclipses H
Stability: Low
✔ Torsional strain present
(b) Fully Eclipsed (CH₃–CH₃)
- Dihedral angle = 0°
- CH₃ groups directly overlap
Stability: Lowest
❌ Maximum torsional strain
❌ Maximum steric repulsion
Energy Order (Important for Exams)