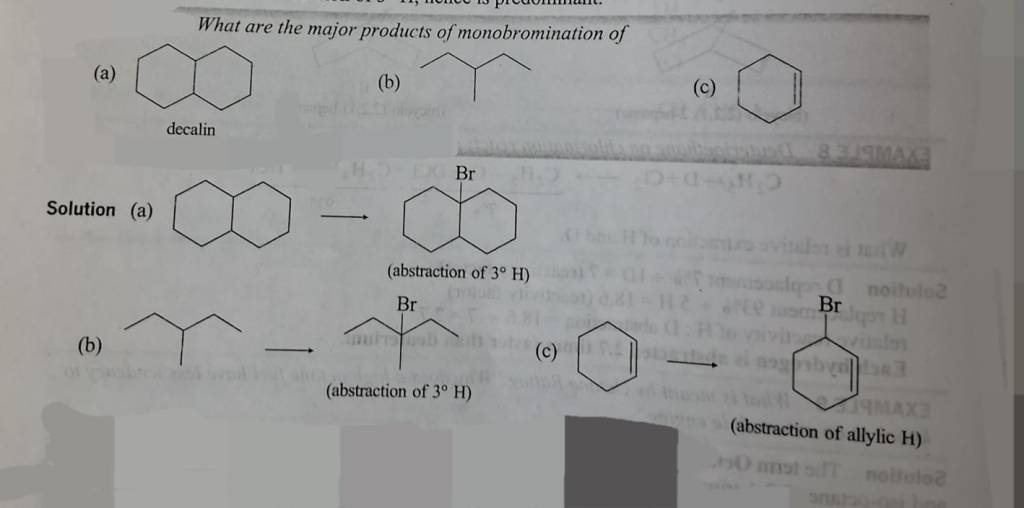
Free radical halogenation is a substitution reaction of alkanes in which a hydrogen atom is replaced by a halogen (Cl or Br) in the presence of UV light (hv) or heat.
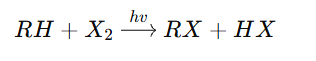
(X = Cl or Br)
Mechanism:
Step 1

Step 2: Propagation (Chain continues)
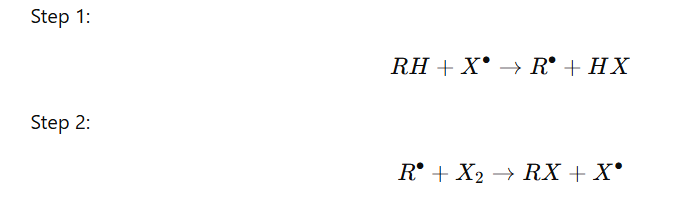
Step 3:Termination (Radicals combine)
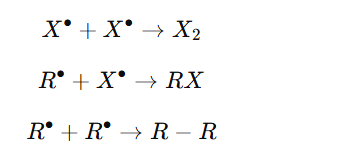
Order of Reactivity of Halogens
But in practice:
- Fluorination → Explosive
- Chlorination → Fast, less selective
- Bromination → Slow, more selective
- Iodination → Not feasible (endothermic)
Selectivity Rule (Very Important for JEE)
Relative Reactivity of Hydrogens
Because:
✔ Stability of radical
✔ Hyperconjugation
✔ Inductive effect
Energy Profile Concept
✔ Chlorination → Low activation difference
✔ Bromination → Large activation difference
👉 Bromination gives major substituted product (Hammond postulate)
JEE / NEET Special Points
✔ Reaction requires UV light
✔ Radical inhibitor (like O₂) slows reaction
✔ Tertiary hydrogen is most reactive
✔ Allylic and benzylic positions are highly reactive
✔ Reaction gives mixture of products
Allylic Halogenation (Very Important Exception)
When alkene is treated with halogen under radical conditions → substitution occurs at allylic position, not addition.
Example:
Propene + Br₂ (hv) → Allylic bromide
Best reagent: NBS (N-Bromosuccinimide)
Reason:
✔ Allylic radical is resonance stabilized
✔ More stable than normal 3° radical sometimes
Benzylic Halogenation
Example:
Toluene + Br₂ (hv) → Benzyl bromide
Reason:
✔ Benzyl radical is highly resonance stabilized
✔ Very reactive position
Reactivity order (special case):
Energy Profile Concept (JEE Advanced Angle)
• Chlorination → Exothermic
• Bromination → Slightly exothermic
• Iodination → Endothermic
Iodination in Presence of HIO₃ (Why It Works?)
Iodination is carried out in presence of HIO₃ because it oxidizes HI to I₂, removing HI and shifting equilibrium forward, making the reaction feasible.
The Problem (Normal Iodination
✔ Propagation step is endothermic
✔ Reaction is reversible
✔ HI formed shifts equilibrium backward
👉 So iodination is normally very slow / does not proceed.
⚡ Role of HIO₃ (Iodic Acid)
When HIO₃ is added:
What happens?
✔ HI (product) is removed
✔ Equilibrium shifts forward (Le Chatelier principle)
✔ Reaction becomes feasible
Stability of Free Radicals (Including Vinylic Radical) – JEE / NEET
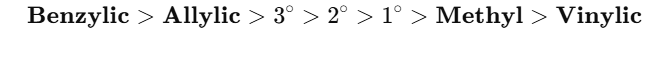
Chlorination – Relative Reactivity of Different Hydrogens (JEE / NEET)
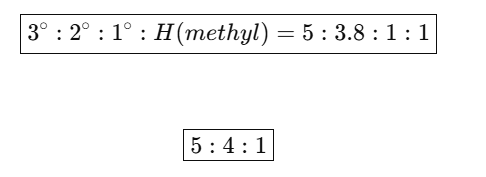
Meaning of the Ratio
- 3° hydrogen reacts about 5 times faster than 1° hydrogen
- 2° hydrogen reacts about 3.8 times faster than 1° hydrogen
- 1° hydrogen ≈ methyl hydrogen (taken as 1)
Why This Order?
Hydrogen abstraction forms a radical:
Since radical stability order is:
More stable radical → lower activation energy → faster reaction.
But remember
Chlorination is less selective, so differences are moderate.
Comparison with Bromination (Very Important for JEE)
For bromination:
👉 Bromination is highly selective.
👉 Chlorination is fast but less selective.
Numerical 1
Calculate percentage of products formed when propane undergoes chlorination.
Step 1: Identify hydrogens
Propane (CH₃–CH₂–CH₃)
- 6 primary H
- 2 secondary H
Step 2: Multiply by reactivity ratio
Primary:
Secondary:
Step 3: Total
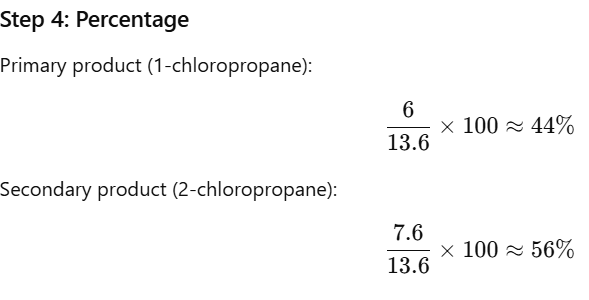
Final Answer
✔ 1-chloropropane ≈ 44%
✔ 2-chloropropane ≈ 56%
Order of C–H Bond Dissociation Energy (BDE)
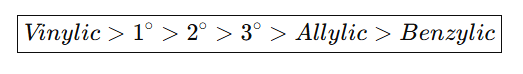
Bond strength decreases as radical stability increases
Question:
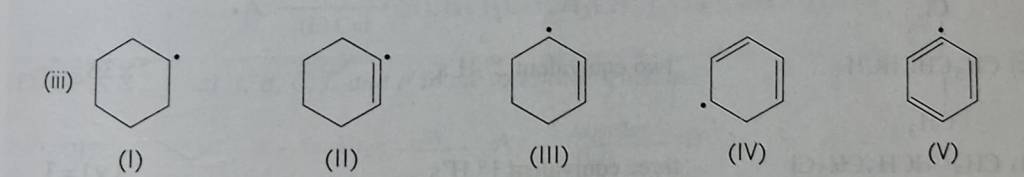
Ans: IV > III> 1 > II >V