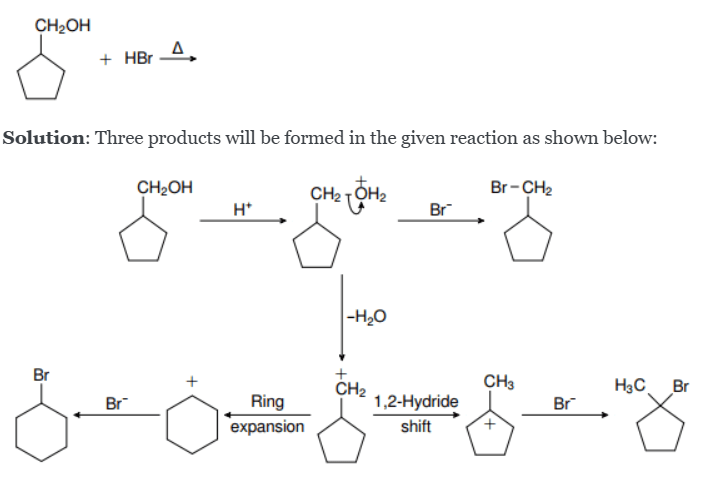
What is Ring Expansion?
Ring expansion is a rearrangement reaction where a cyclic compound increases its ring size (e.g., 5-membered → 6-membered ring).
It usually happens during carbocation rearrangements or nitrene/carbene insertions.
🔹 Why Does It Happen?
Rings expand mainly for stability reasons:
- Carbocation stability – a less stable carbocation shifts to form a more stable one.
- Ring strain relief – small rings (3, 4, 5-membered) are strained; expansion to 6-membered reduces strain.
- Formation of more stable rings – 6-membered rings are especially stable due to minimal angle and torsional strain.
🔹 General Mechanism
Typical situation: a carbocation is adjacent to a ring.
- A bond adjacent to the carbocation migrates (1,2-shift) into the positive center.
- This migration enlarges the ring size by one carbon.
Stepwise:
- Generate a carbocation (often via protonation, halonium ion, or leaving group loss).
- Adjacent C–C bond migrates toward the carbocation center.
- New carbocation forms inside the enlarged ring.
Do ring expansion always to cyclohexane?
Ring expansion does not always stop at cyclohexane, but cyclohexane is the most common and stable product when smaller rings expand, because:
- Cyclohexane is strain-free (ideal bond angles ~109.5°, no angle strain).
- Rings smaller than 6 (3-, 4-, 5-membered) often expand to reach 6.
- Rings larger than 6 (like 7 or 8) may rearrange in different ways but they don’t necessarily “shrink” back to 6.
General trends:
- Cyclopropane (3-membered) → expands to cyclobutane, cyclopentane, or cyclohexane (depending on the mechanism).
- Cyclobutane (4-membered) → prefers expansion to cyclopentane or cyclohexane (to reduce strain).
- Cyclopentane (5-membered) → expands most often to cyclohexane (since 6 is especially stable).
- Cyclohexane (6-membered) → usually does not expand, because it’s already the most stable.
- Cycloheptane or larger → can undergo rearrangements, but not necessarily to cyclohexane; instead, they may give different products depending on the reaction condition
