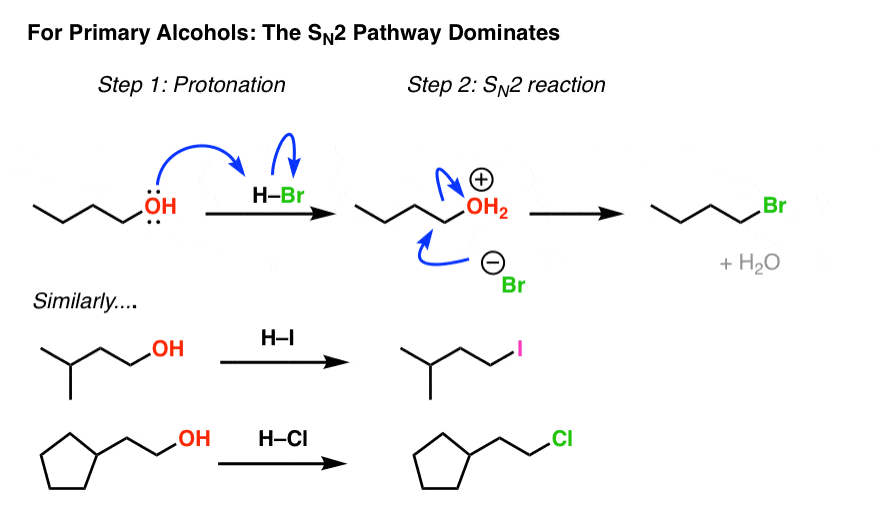
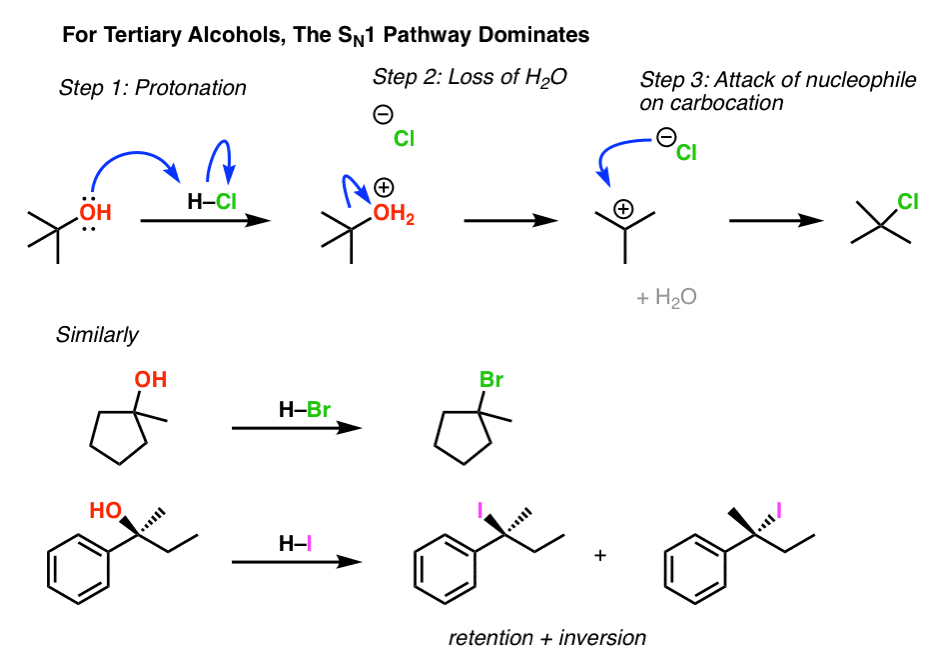
In SN1 (Substitution Nucleophilic Unimolecular) reactions involving alcohols (R-OH), the mechanism requires the departure of a leaving group to form a carbocation intermediate in the rate-determining step. The hydroxyl group (OH) in alcohols is a poor leaving group because it is a strong base (OH⁻) and holds onto the substrate too tightly, making ionization difficult.
To facilitate the reaction, the alcohol is first protonated under acidic conditions (e.g., using H₂SO₄ or HCl) to form an alkyloxonium ion (R-OH₂⁺). This protonation weakens the C-O bond and converts the leaving group to neutral water (H₂O), which is a much better leaving group due to its weaker basicity and greater stability. The water then departs, generating the carbocation (R⁺), which can be attacked by the nucleophile in the subsequent fast step.
Without this protonation, the SN1 pathway would be inefficient or impossible for alcohols, as the energy barrier for OH⁻ departure is too high. This is why SN1 reactions on alcohols are typically conducted in acidic media.