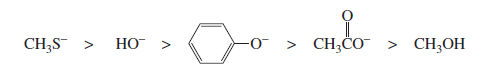
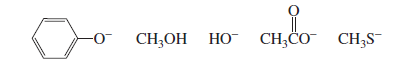Nucleophilicity means the ability of a species to donate an electron pair to an electrophile and form a bond. In exams, students must quickly compare nucleophiles using a few core rules.
When we talk about atoms or molecules that have lone-pair electrons, sometimes we call them
bases and sometimes we call them nucleophiles (Table 9.1). What is the difference between a base
and a nucleophile?
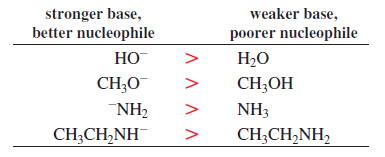
Basicity tells us how easily a compound (base) can donate its lone pair of electrons to a proton (H⁺).
A strong base donates its electron pair more easily than a weak base.
Basicity is related to the acid dissociation constant (Ka) of its conjugate acid, which shows how easily that conjugate acid releases a proton. If the conjugate acid releases H⁺ easily, the base is weaker; if it does not release H⁺ easily, the base is stronger.
Nucleophilicity describes how easily a compound (nucleophile) can attack an electron-deficient atom and form a new bond.
It is measured by the rate constant (k), which indicates how fast the nucleophile reacts in a chemical reaction.
Note: Species with a negative charge is a stronger base and a better nucleophile than a
species that has the same attacking atom but is neutral
If the attacking atoms are the same size, stronger bases are better nucleophiles.
−NH2 > HO− > F−
If, however, the attacking atoms of the nucleophiles are very different in size, another factor
comes into play: the polarizability
Effect of Solvent on Nucleophilicity (Simple Point-wise) 🧪
- Aprotic Polar Solvent
(Example: DMSO, DMF, Acetone)
- Solvent molecules do not contain O–H or N–H bonds.
- There is no strong hydrogen bonding with nucleophiles.
- Therefore nucleophiles remain free and reactive.
- Basicity and nucleophilicity follow the same order.
- Stronger base → stronger nucleophile.
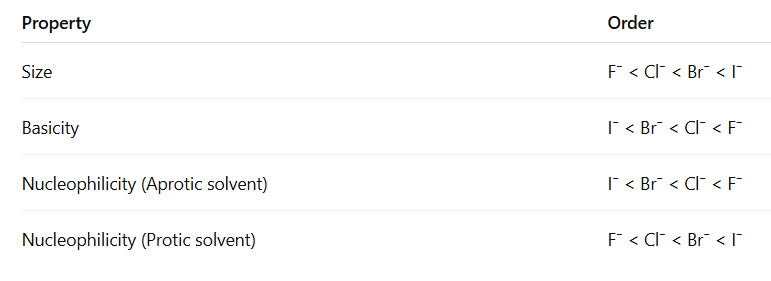
Example (Halide ions) :
- Here I⁻ is the weakest base, so it is the poorest nucleophile in aprotic solvents.
2. Protic Polar Solvent
(Example: Water, Alcohol)
- Solvent molecules contain O–H or N–H bonds.
- They form hydrogen bonding with nucleophiles.
- Small ions get strongly solvated (surrounded by solvent molecules).
- This reduces their nucleophilicity.
Result:
Basicity and nucleophilicity order becomes opposite.
Example (Halide ions):
- I⁻ is the best nucleophile in protic solvents because it is large and less solvated.
✅ Quick NEET/JEE Rule
- Aprotic solvent: Basicity order = Nucleophilicity order
- Protic solvent: Large atom = Better nucleophile
PROBLEM : Indicate whether each of the following solvents is protic or aprotic: a. chloroform (CHCl3) b. diethyl ether c. acetic acid d. hexane
To classify protic vs aprotic solvents, remember the rule:
Protic solvent: contains H bonded to O, N, or F → can form hydrogen bonding and donate H⁺.
Aprotic solvent: no H attached to O, N, or F.
(a) Chloroform
Structure: CHCl₃
- Hydrogen is attached to carbon, not O/N/F.
- Cannot donate hydrogen for hydrogen bonding.
Result: Aprotic solvent
(b) Diethyl ether
Structure: C₂H₅–O–C₂H₅
- Oxygen is present, but no O–H bond.
- Hydrogens are attached only to carbon.
Result: Aprotic solvent
(c) Acetic acid
Structure: CH₃–COOH
- Contains O–H bond in the –COOH group.
- Can donate proton and form hydrogen bonding.
Result: Protic solvent
(d) Hexane
Structure: alkane
- Only C–H and C–C bonds.
- No O–H, N–H, or F–H bonds.
Result: Aprotic solvent
Relative Nucleophilicity toward CH3l in Methanol

PROBLEM: Which is a stronger base: RO– or RS–?
b. Which is a better nucleophile in an aqueous solution?
c. Which is a better nucleophile in DMSO?
Let RO⁻ (alkoxide) and RS⁻ (thiolate) be compared.
Key idea: Basicity and nucleophilicity are not always the same, especially depending on the solvent.
(a) Which is a stronger base?
Compare conjugate acids:
- RO⁻ → conjugate acid Alcohol (pKa ≈ 16)
- RS⁻ → conjugate acid Thiol (pKa ≈ 10)
Lower pKa → stronger acid → weaker conjugate base.
Since thiols are more acidic than alcohols, their conjugate base RS⁻ is weaker.
Stronger base: RO⁻
(b) Better nucleophile in aqueous solution (polar protic solvent)
Water is a protic solvent, so small anions are strongly solvated by hydrogen bonding.
- RO⁻ (O is smaller) → strongly solvated → nucleophilicity decreases
- RS⁻ (S is larger, more polarizable) → less solvated → more reactive
Better nucleophile in water: RS⁻
(c) Better nucleophile in Dimethyl sulfoxide (polar aprotic solvent)
In polar aprotic solvents, anions are not strongly solvated, so nucleophilicity follows basicity.
Since:
RO⁻ is a stronger base than RS⁻
Better nucleophile in DMSO: RO⁻
Which is stronger nucleophile?
(a) Br⁻ or Cl⁻ in H₂O
Water strongly solvates smaller ions.
Br⁻ is larger → less solvated → more reactive.
Better nucleophile: Br⁻
b. CH₃O⁻ or CH₃OH in H₂O
Charged species are stronger nucleophiles than neutral molecules.
Methoxide ion vs Methanol
Better nucleophile: CH₃O⁻
c. CH₃O⁻ or CH₃OH in DMSO
Same logic: anion >> neutral molecule.
Better nucleophile: CH₃O⁻
(d) Br⁻ or Cl⁻ in DMSO
In aprotic solvent → nucleophilicity follows basicity.
Basicity order:
Cl⁻ > Br⁻
Better nucleophile: Cl⁻
(e) HO⁻ or NH₂⁻ in H₂O
Hydroxide ion vs Amide ion
NH₂⁻ is a much stronger base, but in water it reacts with the solvent:
NH₂⁻ + H₂O → NH₃ + HO⁻
So NH₂⁻ cannot exist effectively in water.
Better nucleophile: HO⁻
(f) I⁻ or Br⁻ in H₂O
In protic solvent → larger ion is better nucleophile.
I⁻ > Br⁻
Better nucleophile: I⁻
(g) I⁻ or Br⁻ in DMSO
Aprotic solvent → nucleophilicity follows basicity.
Basicity:
Br⁻ > I⁻
Better nucleophile: Br⁻
(h) HO⁻ or NH₂⁻ in DMSO
DMSO is aprotic → nucleophilicity follows basicity.
NH₂⁻ is stronger base.
Better nucleophile: NH₂⁻
Nucleophilicity Is Affected by Steric Effects
Basicity Comparison
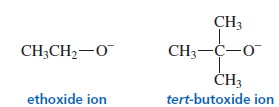
tert-Butoxide (t-BuO⁻) has three methyl groups pushing electron density toward oxygen.
This increases electron density, making it a stronger base.
Stronger base:
Nucleophilicity Comparison
Nucleophilicity depends strongly on steric hindrance.
- tert-Butoxide ion is bulky because of three CH₃ groups.
- This blocks approach to the electrophilic carbon in SN2 reactions.
Ethoxide is smaller, so it can attack carbon more easily.
PROBLEM :
Rank the following species from best nucleophile to poorest nucleophile in an aqueous solution: