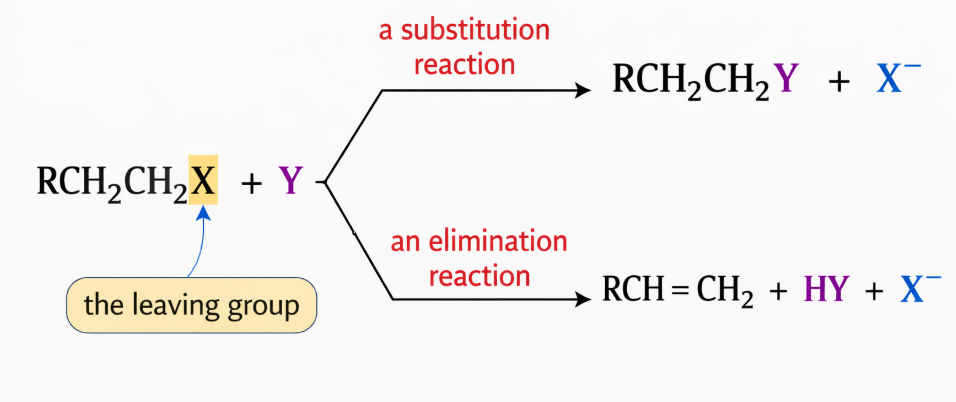
SN2 stands for:
S → Substitution
N → Nucleophilic
2 → Bimolecular
So the full meaning is:
SN2 = Substitution Nucleophilic Bimolecular reaction
Key Features of SN2 Reaction
- Bimolecular Reaction
- Rate depends on two species: substrate and nucleophile.
- Second-Order Kinetics
- Rate law:
- One-Step Mechanism (Concerted Reaction)
- Bond formation and bond breaking occur simultaneously.
- No intermediate is formed.
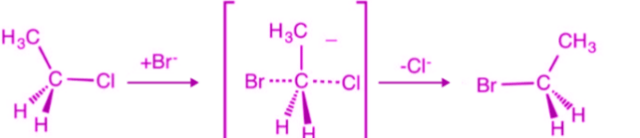
- Backside Attack
- Nucleophile attacks from the opposite side of the leaving group.
- Walden Inversion (Inversion of Configuration)
- If the carbon is chiral, configuration inverts (like umbrella flipping).
- Transition State Formation
- A pentacoordinate transition state is formed.
- Substrate Reactivity Order
- Tertiary halides do not undergo SN2 due to steric hindrance.
- Strong Nucleophile Required
- Examples:
OH⁻, CN⁻, I⁻, RS⁻
- Examples:
- Polar Aprotic Solvents Favor SN2
- Examples: DMSO, DMF, acetone
- They increase nucleophilicity.
- Good Leaving Group Needed
- Order:
- Steric Hindrance Slows Reaction
- Bulky groups near the reacting carbon reduce SN2 rate.
Steric hindrance decreases the reaction rate
- As the alkyl group becomes bulkier, the rate of SN2 decreases.
- This happens because the nucleophile attacks from the backside, and bulky groups block this attack.

An example of inversion:
PROBLEM :
Which substitution reaction takes place more rapidly?
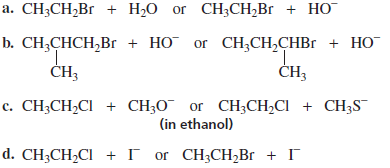
Use SN2 rules to compare the rates:
Key ideas for SN2 (JEE/NEET):
- Stronger nucleophile → faster
- Less steric hindrance → faster
- Better leaving group → faster
- Polar aprotic solvent increases nucleophilicity
Answers
(a)
CH₃CH₂Br + H₂O
or
CH₃CH₂Br + HO⁻
HO⁻ is a much stronger nucleophile than H₂O.
Faster:
CH₃CH₂Br + HO⁻
(b)
(CH₃)₂CHCH₂Br + HO⁻
or
(CH₃)₃CBr + HO⁻
First = primary bromide
Second = tertiary bromide
SN2 order:
Primary > Secondary >> Tertiary
Tertiary halides do not undergo SN2 easily.
Faster:
(CH₃)₂CHCH₂Br + HO⁻
(c)
CH₃CH₂Cl + CH₃O⁻ (in ethanol)
or
CH₃CH₂Cl + CH₃S⁻
Sulfur nucleophiles are stronger than oxygen nucleophiles because they are more polarizable.
Nucleophilicity:
RS⁻ > RO⁻
Faster:
CH₃CH₂Cl + CH₃S⁻
(d)
CH₃CH₂Cl + I⁻
or
CH₃CH₂Br + I⁻
Better leaving group → faster reaction.
Leaving group ability:
Br > Cl
Faster:
CH₃CH₂Br + I⁻
Final Answers
a) CH₃CH₂Br + HO⁻
b) (CH₃)₂CHCH₂Br + HO⁻
c) CH₃CH₂Cl + CH₃S⁻
d) CH₃CH₂Br + I⁻
SN1 stands for:
S → Substitution
N → Nucleophilic
1 → Unimolecular
So the full meaning is:
SN1 = Substitution Nucleophilic Unimolecular reaction
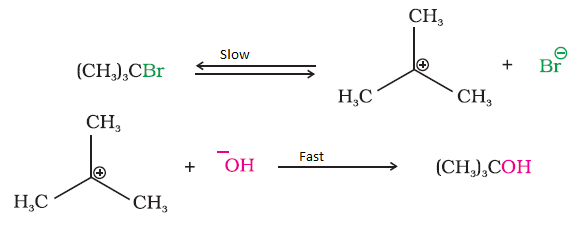
Features of SN1 Reaction (JEE / NEET level)
- Unimolecular Reaction
- Rate depends only on alkyl halide (substrate).
- First-Order Kinetics
- Two-Step Mechanism
- Step 1: Formation of carbocation (slow, rate-determining)
- Step 2: Nucleophile attack (fast)
- Carbocation Intermediate Forms
- Stability of carbocation controls reaction rate.
- Reactivity Order of Substrate
- Polar Protic Solvents Favor SN1
- Examples: H₂O, ROH, ethanol
- Weak Nucleophile is Sufficient
- Examples: H₂O, ROH
- Carbocation Rearrangement Possible
- Hydride shift or methyl shift may occur.
- Racemization of Product
- If the carbon is chiral, product becomes racemic mixture.
- Good Leaving Group Required

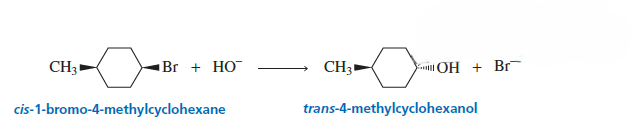
Most SN1 reactions give partial racemization.
In an SN1 reaction, a carbocation intermediate is formed, which is planar. Because of this planar structure, the nucleophile can attack from both sides, leading to formation of two stereoisomers.
However, in most cases the products are not formed in equal amounts. Usually, 50–70% of the product has inverted configuration, while the rest has retained configuration.
If equal amounts of both stereoisomers are produced, the reaction is called complete racemization.
When more of the inverted product is formed, the reaction is called partial racemization
Factors Affecting SN1 Reactions
The rate of an SN1 reaction is influenced mainly by the leaving group and the nucleophile.
Effect of Leaving Group in SN1 Reaction
In an SN1 reaction, the rate-determining step is the formation of a carbocation. Therefore, the reaction rate depends on two important factors:
- Ease of leaving group departure
- The reaction is faster when the leaving group can easily dissociate from the substrate.
- Stability of the carbocation formed
- The more stable the carbocation, the faster the SN1 reaction occurs.
Therefore, good leaving groups and stable carbocations increase the rate of SN1 reactions.

PROBLEM :
Which of the following reactions take place more rapidly when the concentration of the nucleophile is increased?
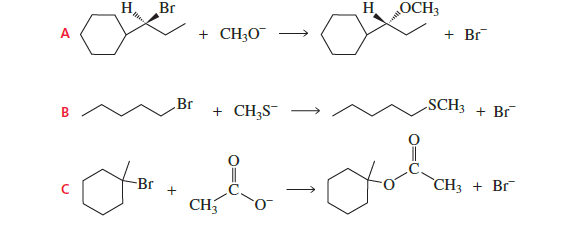
he question asks: Which reaction becomes faster when the nucleophile concentration increases?
Key concept:
- SN2 reaction: Rate = k[R–X][Nu−] → depends on nucleophile concentration
- SN1 reaction: Rate = k[R–X] → independent of nucleophile concentration
So we must identify which reactions proceed by SN2.
Reaction A
Substrate: secondary alkyl bromide attached to cyclohexane
This structure favors SN1 (carbocation formation possible).
Therefore rate does NOT depend strongly on nucleophile concentration.
❌ Not the answer
Reaction B
Substrate: primary alkyl bromide
Primary halides undergo SN2 easily.
Rate depends on nucleophile concentration (CH₃S⁻).
Correct
Reaction C
Substrate: tertiary bromide on cyclohexane
Tertiary halides react by SN1.
Rate depends only on substrate, not nucleophile.
❌ Not the answer
