Isomerisation of alkane is the process in which a straight-chain alkane (n-alkane) is converted into its branched-chain isomer without changing the molecular formula.
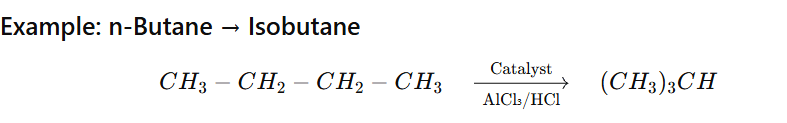
Conditions Required
- Catalyst:
- Anhydrous AlCl₃
- HF
- Pt/Al₂O₃
- Temperature: 250–400°C (industrial process)
- Occurs via carbocation mechanism
Mechanism (Simplified for JEE/NEET)
- Formation of carbocation
- Hydride shift or methyl shift
- Formation of more stable branched carbocation
- Deprotonation → Branched alkane
Industrial Importance
- Used in petroleum refining
- Improves octane number of petrol
- Branched alkanes burn more smoothly (less knocking)
Example:
n-Pentane → Isopentane
n-Hexane → Isohexane
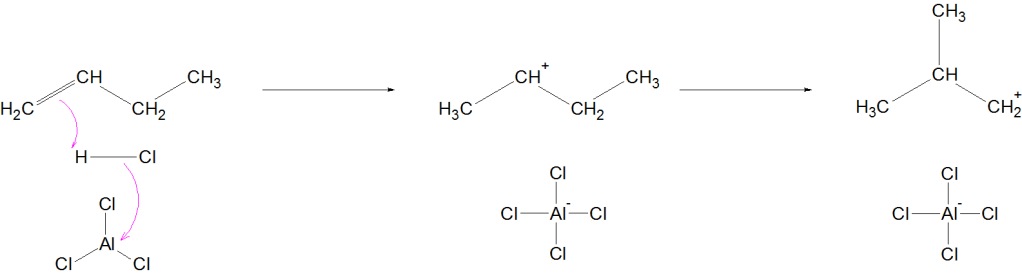
The mechanism for butane:
it probably begins by the protonation of butene impurities.
Step 1: Initiation
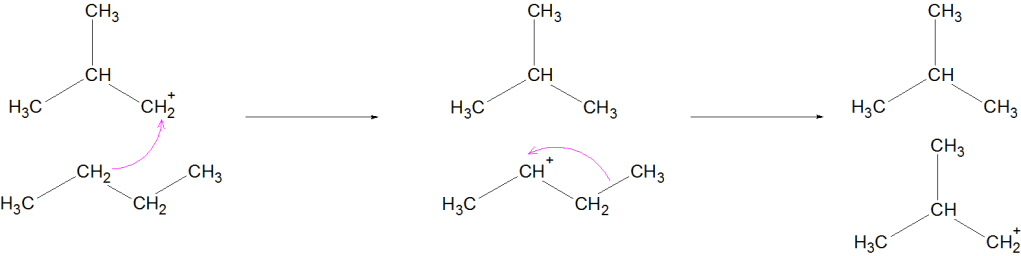
Propagation:
Termination:
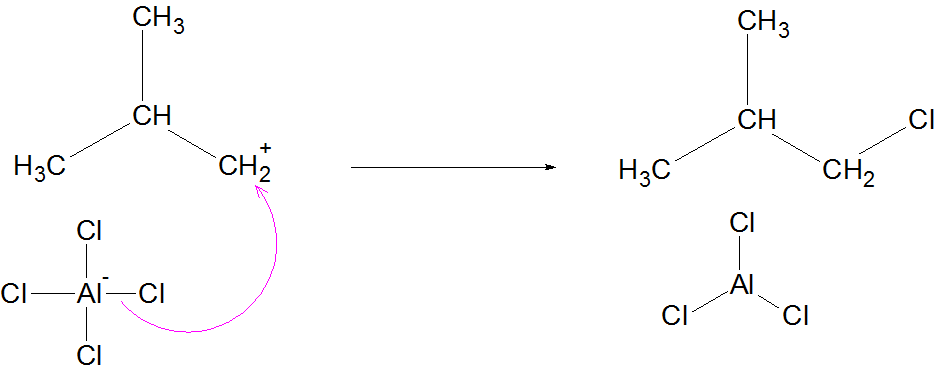
Isomerisation of Pentane (C₅H₁₂)
During catalytic isomerisation of n-pentane (using AlCl₃/HCl or Pt/Al₂O₃), rearrangement proceeds via a carbocation intermediate.
🔹 Possible products:
2,2-Dimethylpropane (Neopentane)
2-Methylbutane (Isopentane)
Major Product → 2-Methylbutane (Isopentane)
✔ Why is it major?
- Formation occurs via secondary carbocation, which is reasonably stable.
- It requires only one methyl shift from n-pentane.
- Formation of neopentane requires further rearrangement and is less favored.
- Under equilibrium, moderately branched alkane forms in higher amount.
Isomerisation of n-Hexane — Major Product (Isohexane)
When n-hexane (C₆H₁₄) undergoes catalytic isomerisation (AlCl₃/HCl, Pt/Al₂O₃, zeolites), branched isomers are formed via carbocation rearrangement.
🔹 Possible Isohexanes (Structural Isomers)
1) 2-Methylpentane ✅ Major Product
2) 3-Methylpentane
3) 2,2-Dimethylbutane
4) 2,3-Dimethylbutane
✅ Major Isohexane → 2-Methylpentane
✔ Reason (JEE/NEET Concept):
- Forms via secondary carbocation with minimal rearrangement.
- Requires only one hydride/methyl shift.
- Moderately branched alkane is favored kinetically.
- Highly branched (like 2,2-dimethylbutane) needs further rearrangement → less formed.
Practically (Industrial Catalytic Isomerisation)
- Commercially important for C₄ to C₇/C₈ alkanes
- Used in petroleum refining to increase octane number.
- Higher alkanes (> C₈) tend to undergo cracking along with isomerisation.
