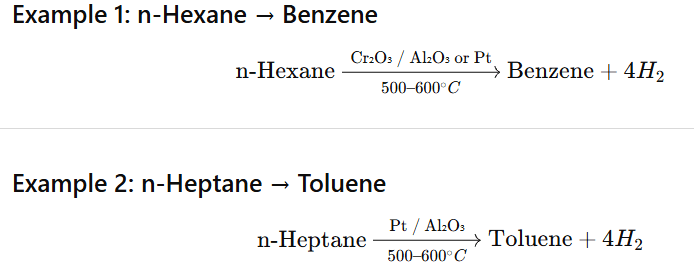
Aromatisation is the conversion of higher alkanes (C₆ or more) into aromatic hydrocarbons (benzene and its derivatives) by dehydrogenation and cyclisation at high temperature in presence of catalysts.
n-Octane (C₈) → Xylene (or ethylbenzene)
Also possible under catalytic reforming conditions.
Theoretical Point of View
There is no fixed maximum carbon limit.
Any C₆ or higher straight-chain alkane (C₆⁺) can undergo aromatisation because at least six carbons are required to form a benzene ring.
🔹 Practical / Industrial Point of View
In petroleum refining (catalytic reforming):
✔ Most effective for C₆ to C₁₀ alkanes
✔ Above C₁₀–C₁₂, cracking and side reactions increase
✔ Very long chains prefer cracking rather than clean aromatisation
So practically:
Cyclohexane undergoes multiple dehydrogenations:
👉 Total 4 molecules of H₂ released.
🔹 Key Concept
✔ First dehydrogenation, then cyclisation, then aromatic stabilization
✔ Metal catalyst helps in removal of hydrogen
✔ Final product gains extra stability due to aromatic resonance
