Chemistry Notes of Structure of Atom
Cathode ray discharge tube experiment/Discovery Of Electron.
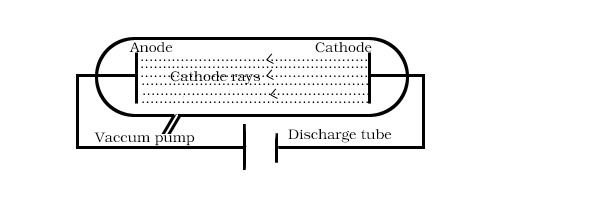
When high voltage is applied to discharge tube at very low pressure ,current starts flowing through a stream of particles moving in the tube from –ve electrode(Cathode) to the +ve electrode(anode).These were called cathode rays particle latter these particles were called electrons.
What are the properties of cathode rays?
1.The cathode rays move towards anode from cathode.
2.They travel in straight line.
3.Deviations of cathode rays in electric field and magnetic field are similar to that the cathode rays consists of –vely charged particles.
4.Properties of cathode rays(such as e/m ratio) do not depend upon the materials of electrode and the nature of the gas present in the cathode ray tube.
How protons were discovered? What are canal rays?
Goldstein used a gas discharge tube which had a perforated cathode.When a high electrical potential is applied between the cathode and anode,”rays” are seen extending from the holes in the back of the cathode. These rays are beams of particles moving in a direction opposite to the cathode rays which are streams of electrons which move toward the anode. “channel rays” or “canal rays”, because they were produced by the holes or channels in the cathode. The lightest +ve ions, formed when there was some hydrogen gas in the tube .They were called protons.
What are the properties of anode rays?
1.Properties of anode rays (e/m ratio etc,) depend upon the gas present in the discharge tube.
2.Anode rays are +vely charged gaseous ions.
3.Properties of anode rays in electrical and magnetic field are opposite to the properties of cathode rays.
How neutrons were discovered?
Chadwick bombarded a thin sheet of Be by α-particles, new types were emitted.He named these particles neutrons.Neutrons have mass slightly greater than of protons and have no charge.
Thomson model of atom or plum pudding or watermelon model of atom.
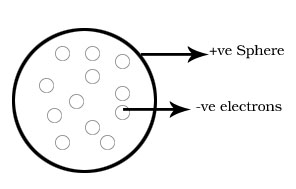
1.Atom has spherical shape.
2.Positive charge is uniformly distributed throughout the atom.
3.The electrons are embedded into it in such a way which gives the most stable electrostatic arrangement.
4.Atom has no charge.
Explain Rutherford Model of atom.
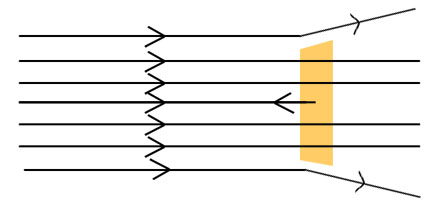
The thin foil of gold was bombarded by α-particle, the following observations were made:
(i) Most of the fast moving α- particles passed straight through the gold foil.
(ii) Some of the α- particles were deflected by the foil by small angles.
(iii) Surprisingly one out of every 12000 particles appeared to rebound.
Rutherford concluded from the α particles scattering experiment that–
(i) Most of the space inside the atom is empty because most of the α-particles passed through the gold foil without getting deflected.
(ii) Very few particles were deflected from their path, indicating that the positive charge of the atom occupies very little space.
(iii) A very small fraction of α-particles were deflected by 180o, indicating that all the positive charge and mass of the gold atom were concentrated in a very small volume within the atom.
On the basis of his experiment, Rutherford put forward the nuclear model of an atom, which had the following features:
(i) There is a positively charged center in an atom called the nucleus. Nearly all the mass of an atom resides in the nucleus.
(ii) The electrons revolve around the nucleus in well-defined orbits.
(iii) The size of the nucleus is very small as compared to the size of the atom.
What is was the drawback of Rutherford atomic model of atom?
The orbital revolution of the electron is not expected to be stable. Any particle in a circular orbit would undergo acceleration. During acceleration, charged particles would radiate energy. Thus, the revolving electron would lose energy and finally fall into the nucleus. If this were so, the atom should be highly unstable and hence matter would not exist in the form that we know. We know that atoms are quite stable.
Atomic number and Mass number.
ATOMIC NUMBER- The number of protons in the nucleus of an atom determines an element’s atomic number.
MASS NUMBER-Number of protons + number of neutrons is called mass number.
Define Isotopes and isobars with examples.
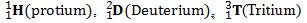
ISOBARS-Atoms with same mass number but different atomic numbers are called mass numbers.
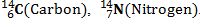
Define electromagnetic waves or electromagnetic radiation.
When electrically charged particle move under acceleration, alternating electrical and magnetic fields are produced and transmitted in the form of waves called electromagnetic wave or electromagnetic radiation.
What are the properties of electromagnetic wave?
1.Oscillating electric and magnetic fields are perpendicular to each other and both are perpendicular to the direction of propagation of the wave.
2.They do not require medium and can move in vacuum.
3.There are many types of electromagnetic wave.
How is the frequency(٧) wavelength(λ), and the velocity(C) of light are related among each other?
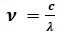
Define wave number.
It is defined as the number of wavelength per unit length.
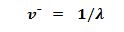
Arrange different types of electromagnetic waves in increasing order of their frequency.
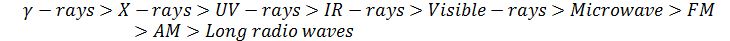
What is black body? What is black body radiation?
The ideal body which emits and absorbs all frequencies is called black body and the radiation emitted by such a body is called black body radiation.
What is the relation between intensity and wavelength for a black body radiation.
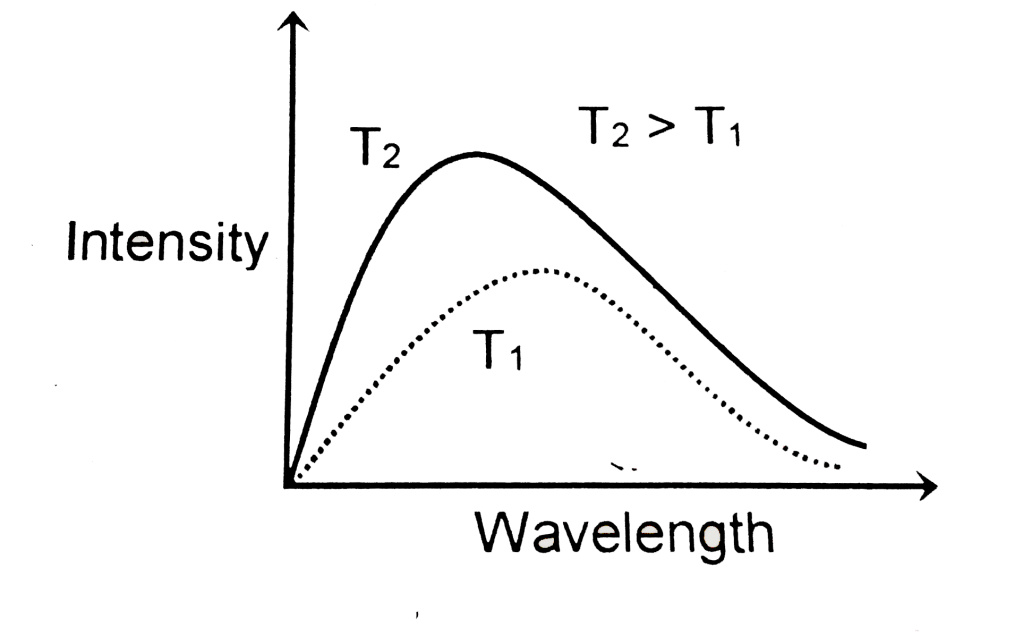
At given temp ,intensity of radiation emitted increases with decreases of wavelength ,reaches at maximum value and the start decreasing with further decrease of wavelength.
Which properties of electromagnetic wave that is not explained by wave properties.
1.Black body radiation.
2. Photoelectric effect.
What is Planck’s quantum theory ?
1.The radiant energy which is emitted or absorbed by the black body is not continuous but discontinuous in the form of small discrete packets of energy, each such packet of energy is called a ‘quantum‘. In case of light, the quantum of energy is called a ‘photon‘.
2.The energy E of the quantum is related to the frequency ![]() .The quantity h, is Planck constant, is a universal constant with the value of 6.626 X 10-34 Js.
.The quantity h, is Planck constant, is a universal constant with the value of 6.626 X 10-34 Js.
What is photoelectric effect?
When certain metals (K.Rb.Cs etc) are exposed by a beam of light electrons are ejected from metals, the phenomena is called photoelectric effect.
Which observation of photoelectric effect explains the particle nature of light?
Ejection of electron depends on the frequency of light and it does not depend on the intensity of light.
Define Work function.
Minimum energy that can eject electron from metal surface.
Define threshold frequency
Minimum frequency below which photoelectric effect is not observed.
Give the relation between energy of photon, work function and kinetic energy during photoelectric effect.
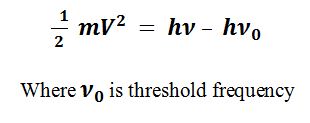
On which factor no. of ejection of electrons depends.
The number of photoelectrons ejected is proportional to the intensity of incident radiation.
How can we say that electromagnetic radiations have dual behavior?
Interference and diffraction phenomena can explain by wave nature.
Photoelectric effect and blackbody radiation can explain by particle nature.
Define spectrum of white light
A ray of white light spread into series of bands called spectrum .Angle deviation is inversely proportional to the wavelength if the wave. Red color deviated the least while violet is deviated the most.
What is continuous spectrum?
A spectrum having no apparent gap or breaks throughout its entire spectrum.
Define emission spectra.
The emission spectra are produced by thin gases in which the atoms do not experience many collisions .The emission lines corresponds the photon of discrete energies that are emitted when excited atomic states in the gas back to the lower states.
Define absorption spectrum.
An absorption spectrum occurs when white light passes through a cold, dilute gas and atoms in the gas absorbs at characteristic frequencies, thus give rise to dark lines in the spectrum.
Define line spectra.
Atoms emit light only a specific wavelength with dark space between them called spectra.
Explain line spectrum of Hydrogen
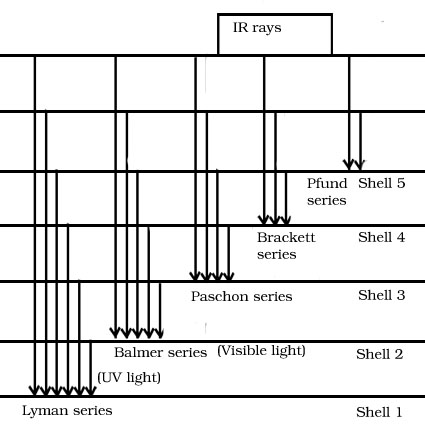
When an electric discharge is passed through gaseous Hydrogen excited Hydrogen atoms produced emit electromagnetic radiation.
Types of different transition in spectrum of Hydrogen.
1.Lyman series-It involves transition which end with the ground state of Hydrogen(n=1)
2.Balmer series-It involves transition which end with shell n=2.
3.Paschon series-It involves transition which end with shell n=3
4.Brackett series-It involves transition which end with shell (n=4)
5..Pfund series-It involves transition which end with the shell (n=5)
The postulates of Bohr’s atomic model.
1.Electrons move around the nucleus in a circular path of fixed radius and energy fixed paths are called orbit.
2.The energy of an electron in the orbit does not change with time.
3.Energy is absorbed when electron move from a lower stationary to a higher stationary state to and energy is emitted when electrons move from higher stationary state to lower state.
4.The frequency of radiation absorb or emitted is equal to
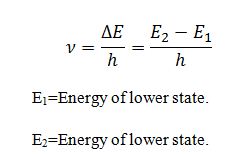
5.Electrons can move only in those orbit for which its angular momentum is integral multiple of h/2π.
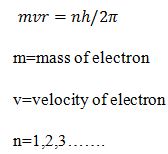
What are the advantages of Bohr’s Model.
1.The radii of the stationary are expressed as rn=52.9 n2/Z pm
2.Energy of stationary state
En = -2.18×10-18 Z2 (1/n2) J.
Z=atomic number
3.Bohr’s theory can also be applied to the ions containing 1 electron example-He+.Li2+,Be3+
4..Velocity of electron moving in orbit can also be calculated.
Formula for calculating energy, frequency released or absorbed in line spectrum of Hydrogen.

What are the limitation of Bohr’s model?
1.It fails to account for the finer detail of the Hydrogen spectrum.
2.Unable to explain the spectrum of atoms other than Hydrogen atom.
3.This model is unable to explain the splitting of spectral lines in presence of magnetic field(Zeeman effect) or an electric field(stark effect).
4.Could not explain the ability of atoms to form molecule.
What is the reason for the failure of the Bohr’s model?
Bohr model of Hydrogen atom not only ignores dual behavior of matter but also contradicts Heisenberg uncertainty principle.
What is quantum mechanics?
The branch of science deals with the dual behavior of matter.
Write the Schrodinger wave equation for a system whose energy does not change with time.
H^=Eψ where H^ is a mathematical operator called Hamiltonian operator.
What do you mean by ψ and ψ2.
Schrodinger equation gives the possible energy levels the electron can occupy and corresponding wave function (ψ).The wave function ψ denotes an atomic orbital for an electron in an atom.Ψ2 is probability density where finding an electron is maximum.
Plot a graph for orbital wave function ψ(r) and the variation of probability density ψ2(r) as a function of distance r of the electron from the nucleus for 1s and 2s orbital.
For 1s orbital the probability density is maximum at the nucleus and it decreases sharply as we move away from it.
For 2s orbital the probability density first decreases sharply to zero and again starts increasing after reaching a small maxima .it decreases again approaches zero as the value of r increases further.
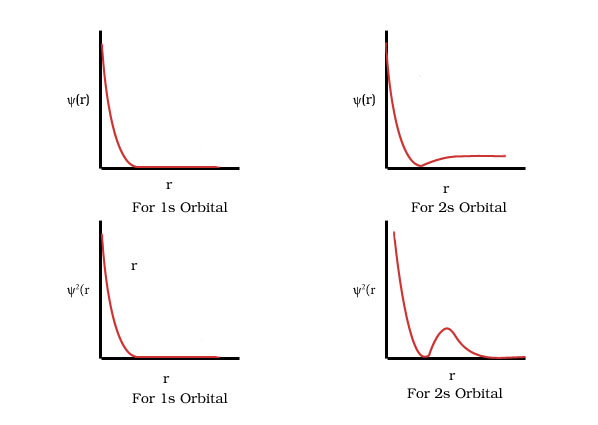
Define nodal surfaces or nodal plane
The region where the probability function reduces to zero is called nodal surfaces.
Give the formula for for determining number of nodes for ns orbital and np orbital.
Ans. For ns orbital number of nodes =(n-1)
For np orbital number of nodes =(n-2)
Give the formula for determining the total number of nodes.
Total number of nodes =angular nodes + radial nodes
=l+(n-l-1)
=n-1
What do you mean by degenerate orbitals?
The orbitals having the same energy are called degenerate orbital.
Explain the de-Broglie theory.
According to de-Broglie theory matter like radiation has both particle and wave like properties.
λ=h/mv=h/P
m=mass of the particle.
V=velocity
P=momentum
Explain the limitations of de-Broglie theory.
de-Broglie theory is not applicable for ordinary objects due to short wavelength is associated with because of large mass of ordinary objects.The wavelengths associated with electrons and other subatomic particles can be detected experimentally.
What is Heisenberg Uncertainty principle.
It is impossible to determine simultaneously the exact momentum and exact position of electron.
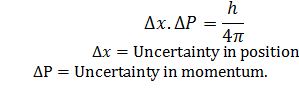
What is the significance of Heisenberg uncertainty principle?
The effect of Heisenberg Uncertainty principle is significant only for motion of microscopic object and is negligible for macroscopic objects.
Let there is a ball have mass 10-6 Kg.

What are Quantum numbers?
Each electron has a set of four quantum numbers called quantum numbers that specify it completely.
Describe the four quantum numbers.
1.Principal quantum numbers-The principal quantum number has the symbol ‘n’where n=1,2,3,4……..it tells which energy level an electron is in .Size and the energy of an orbit can also be determined after the finding the value of n.
2.Azimuthal quantum numbers or subsidiary quantum numbers-The Azimuthal quamtum number determines the orbital angular momentum of an atomic orbital and describes the shape of the orbital.
Value of l depends on n. l can have n value ranging from 0 to (n-1)
The various orbitals related to different values of l are sometimes called subshell.
| symbol | Maximum electron | shape | Name | |
| 0 | s | 2 | sphere | sharp |
| 1 | p | 6 | Two dumbells | principal |
| 2 | d | 10 | 4 dumbells | diffuse |
| 3 | f | 14 | 8 dumbells | fundamental |
3.Magnetic quantum numbers(m)-It describe the orientation in space of a particular orbital.
Value of m depends on value of l. For any possible value of l the values of m are -l to +l
Total value of m= 2l+1
| Subshell | l | m | Possible orientation |
| s | 0 | 0 | Only one way in which a sphere can be oriented in a space |
| p | 1 | -1,0,+1 | Px, py, pz |
| d | 2 | -2,-1,0,1,2 | dxy,dyz,dzx,dx2,dy2,dz2 |
4.Spin quantum number.
An electron spin around its own axis. The two orientation can be describe by spin quantum number and take the values of +1/2 and -1/2.
Draw the shape of s orbital.

Draw the shape of p orbitals.
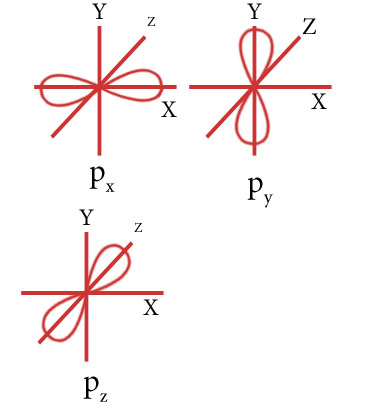
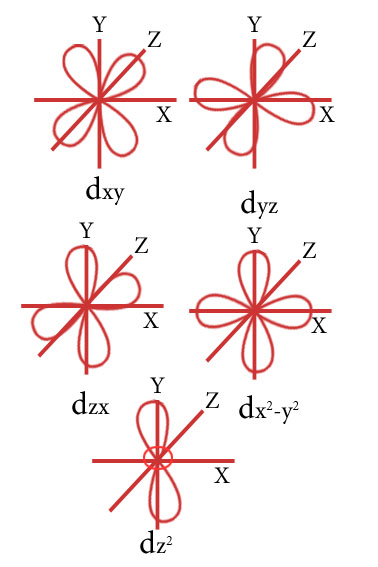
Draw the shape of d orbitals.
What do you mean by valance electron and core electron?
Valence electron-The electrons present in the last shell of atom are called valence electrons.
Core electron-The electron present in completely filled shells are called core electrons.
What is Aufbau principle?
I.Electrons are filled in a orbital in order of their increasing energies.
II.The lower the value (n+l )the lower will be energy of that orbital.
III.If the two orbitals having same (n+l ) the lower the value of n the lower will be energy of that orbitals.
Thus 1s<2s<2p<3s<3p<4s<3d<4p<5s<4d<5p<4f<5d<6p<7s.
What is Pauli exclusion principle?
No two electrons in an atom can have the same set of four quantum numbers.
Which quantum number differentiate the behavior of two electrons?
Two electrons have the same value of n,l,m but must have the opposite spin quantum numbers.
Which rule explain that the maximum number of electrons in the shell with quantum number n is equal to 2n2 ?
Pauli exclusion principle
Explain Hund’s rule
The pairing of electrons in the orbitals belonging to same subshell does not take place until each orbital belonging to that subshell has got one electron.
Write the electronic configuration of Cr.and Cu.
24Cr-1s22s22p63s23p64s13d5.
29Cu-1s22s22p63s23p64s13d10.
Why Cr and Cu adopt d5 and d10 electronic configuration?
Because half filled and fully filled orbitals are more stable.
