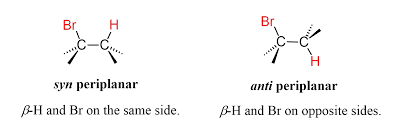In organic chemistry, competition between E1 Reaction and E2 Reaction occurs when a substrate and reaction conditions allow both elimination pathways. The dominant mechanism depends on several factors.
Primary and secondary alkyl halides undergo elimination mainly through the E2 mechanism because the carbocations that would form in an E1 Reaction are relatively unstable, and solvolysis reactions usually involve a high concentration of base that favors the E2 Reaction.
However, tertiary alkyl halides can undergo both E2 and E1 reactions because they are capable of forming relatively stable tertiary carbocations.
Because tertiary alkyl halides are able to undergo both E2 and E1 elimination reactions, the overall rate of the reaction depends on both pathways. Therefore, the rate law is the sum of the rate laws for the E2 and E1 reactions.
Thus, An E2 reaction is favored by a high concentration of a strong base.
An E1 reaction is favored by a low concentration of a weak base
For each of the following reactions, (1) decide whether an E2 or an E1 occurs, and (2) draw the major
elimination product:
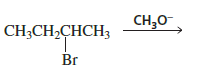
When 2‑Bromobutane reacts with Methoxide Ion, the reaction mainly proceeds through an E2 Reaction because:
- 2-Bromobutane is a secondary alkyl halide.
- CH₃O⁻ (methoxide) is a strong base.
- Strong bases favor E2 elimination.
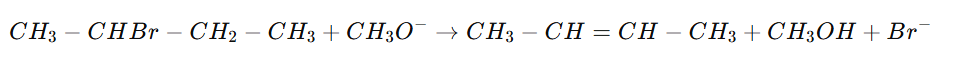
The elimination forms bu-2-tene:
- Major product: 2‑Butene (especially trans-2-butene, most stable)
- Minor product: 1‑Butene
Reason (Zaitsev rule)
According to Zaitsev Rule, the more substituted alkene forms as the major product.
Order of products:
Substrate: secondary alkyl halide
Base: strong (CH₃O⁻)
Mechanism: E2 elimination
Major product: trans-2-butene
Problem: For each of the following reactions, (1) decide whether an E2 or an E1 occurs, and (2) draw the major
elimination product:
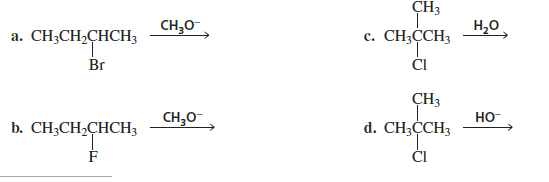

- Strong base → E2
- Weak base + tertiary substrate → E1
- Zaitsev alkene = major product
let’s analyze each reaction using two steps:
Decide E1 or E2
Write the major elimination product
(a)
Substrate: 2-Bromobutane
Reagent: CH₃O⁻ (methoxide) → strong base
Reasoning
- Secondary alkyl halide
- Strong base present
E2 mechanism dominates
Elimination (Zaitsev rule)
The base removes a β-hydrogen giving the more substituted alkene.
Major product
- 2-Butene (mainly trans)
Reaction:
CH₃CH₂CH(Br)CH₃ + CH₃O⁻
→ CH₃CH=CHCH₃ + Br⁻ + CH₃OH
Major alkene:
trans-2-butene
(b)
Substrate: 2-Fluorobutane
Reagent: CH₃O⁻
Reasoning
- Strong base → favors E2
- Fluorine is a poor leaving group, so reaction is slower
- But mechanism is still E2
Major product
Again Zaitsev alkene
Product
2-Butene (trans major)
CH₃CH₂CH(F)CH₃
→ CH₃CH=CHCH₃
(c)
Substrate: tert-Butyl chloride
Structure: (CH₃)₃C–Cl
Reagent: H₂O (weak base, polar protic)
Reasoning
- Tertiary carbocation easily forms
- Weak base
E1 mechanism
Steps:
- Carbocation formation
- Deprotonation
Product
2-Methylpropene (isobutene)
Reaction:
(CH₃)₃CCl → (CH₃)₂C=CH₂
(d)
Substrate: tert-Butyl chloride
Reagent: HO⁻ (strong base)
Reasoning
- Strong base present
- Tertiary substrate
E2 dominates
Product
2-Methylpropene
(CH₃)₃CCl + OH⁻
→ (CH₃)₂C=CH₂
E2 AND E1 REACTIONS ARE STEREOSELECTIVE
Yes, both E2 and E1 elimination reactions are stereoselective, but the degree and reason are different.
1. E2 Reactions – Highly Stereoselective
E2 reactions require a specific geometric arrangement.
Requirement
The β-hydrogen and leaving group must be anti-periplanar (180° apart).
Because of this requirement, the reaction selectively forms the more stable alkene stereoisomer.
Usually:
- Trans (E) alkene is formed more than cis (Z).
Example
2-Bromobutane + strong base
Major product:
- trans-2-butene
Reason:
- Trans alkene is more stable due to less steric repulsion.
Key points:
- Anti-periplanar geometry required
- Reaction is stereospecific and stereoselective
2. E1 Reactions – Moderately Stereoselective
E1 occurs through a carbocation intermediate.
Steps:
- Leaving group leaves → carbocation forms
- Base removes β-hydrogen → alkene forms
Since the carbocation is planar, the base can remove hydrogen from either side.
Result:
- Both cis and trans alkenes may form.
However:
Trans alkene is usually major because it is thermodynamically more stable.
Example
tert-Butyl chloride + H₂O
Product:
- 2-methylpropene
(Only one alkene possible here)