How are alkyl halides reduced?
Alkyl halides can be reduced to alkanes by replacing the halogen atom with a hydrogen atom. This reduction typically involves the use of reducing agents.
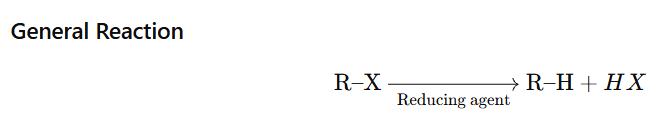
Alkyl halides are reduced to alkanes by:
- Nascent hydrogen (Zn/HCl)
- Zn/CH3COOH
- Zn + NaOH
- LiAlH₄
- Catalytic hydrogenation (H₂/Pd or Ni)
- Zn- Cu Couple +C2H5OH
- TPH (Triphenylhydride / Triphenyltin Hydride)
- Via Grignard reagent followed by hydrolysis
Important Note for JEE/NEET
- Reduction with Nascent Hydrogen Common in laboratory conditions.
- Reduction with Hydrogen (Catalytic Hydrogenation)
- Using H₂ with Pd/C, Ni or Pt catalyst , Effective especially for benzyl and allyl halides.
- LiAlH₄ (Lithium aluminium hydride) Very effective for primary and secondary halides not for 3 degree.With LiAlH₄
- Hydride (H⁻) acts as a strong nucleophile. Mechanism: Direct SN2 displacement of X⁻ by H⁻
- NaBH4 and TPH are used to reduce 3 degree haloalkane to alkane.
- Zn–Cu couple reduces alkyl halides to alkanes (especially in alcohol medium). Zn–Cu More Reactive than Zn? Copper coating removes oxide layer from zinc, increases surface activity , facilitates electron transfer, acts as better reducing system.
- Reduction of Alkyl Halides by TPH. Reaction proceeds via free radical mechanism

