Cracking = breaking of C–C bonds of higher alkanes to give smaller alkanes + alkenes.
Types of Cracking
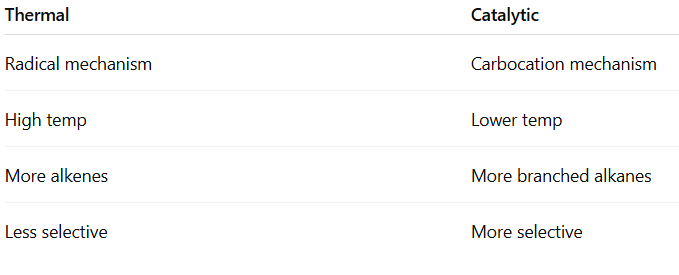
(A) Thermal Cracking
- High temperature (700–900°C)
- Free radical mechanism
- Produces more alkenes
- Less selective
(B) Catalytic Cracking
- 450–550°C
- Zeolite (acidic catalyst)
- Carbocation mechanism
- More branched alkanes
- Higher octane number
Thermal Cracking Example (Free Radical)
Example 1:
Example 2:
Major product contains more substituted alkene due to radical stability
Example 3:
Catalytic Cracking Example (Carbocation Mechanism)
Occurs in presence of zeolite catalyst (acidic sites).
✔ Rearrangement possible
✔ Branched products favored
Example 5:
More stable tertiary carbocation pathway preferred.
