R/S configuration is used to describe the absolute configuration of a chiral carbon (asymmetric carbon with 4 different groups)
Step-by-Step Rules (Cahn–Ingold–Prelog Rules)
Step 1: Assign Priority (1 → 4)
- Higher atomic number = higher priority
- Example:
I > Br > Cl > S > O > N > C > H
If same atom, move to next atoms (tie-breaker rule)
Step 2: Orient the Molecule
- Keep the lowest priority group (4) away from you (behind the plane)
Step 3: Trace Path (1 → 2 → 3)
- Clockwise direction → R (Rectus)
- Anticlockwise direction → S (Sinister)

Important Shortcut
If the lowest priority (4) is towards you (front):
- Reverse the result
- Clockwise → S
- Anticlockwise → R
Key Points for Exams
- R/S is about 3D arrangement, not optical rotation (+/−)
- Always check lowest priority position
- Double/triple bonds are treated as multiple single bonds
Quick Trick (Exam Speed)
“4 behind → direct result“4 in front → reverse result”
The R and S configuration for each stereogenic centre in this from top to bottom is:
A.R, R, R
B.R, S, S
C.R, S, R
D.S, S, R
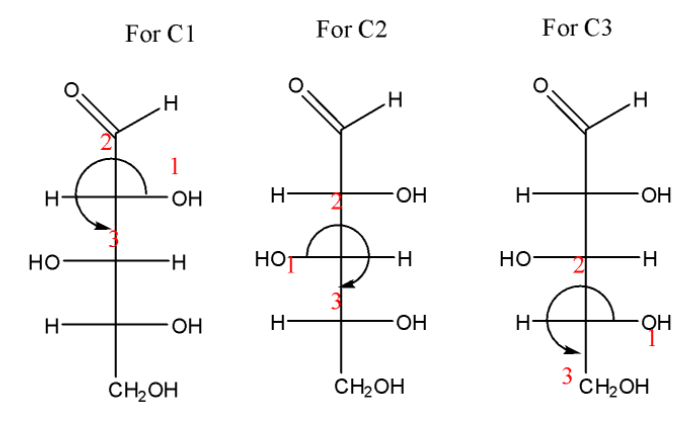
The correct option is (C)
The R and S Configuration Practice Problems
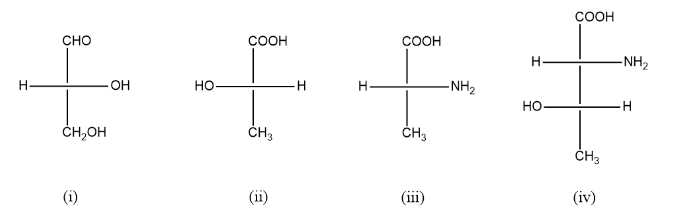
(i)
Groups: OH, CHO, CH₂OH, H
Priority: OH (1) > CHO (2) > CH₂OH (3) > H (4)
- H is horizontal (front) → ❗reverse result
- 1 → 2 → 3 = anticlockwise → reversed → R
✔️ Answer: (i) → R
(ii)
Groups: OH, COOH, CH₃, H
Priority: OH (1) > COOH (2) > CH₃ (3) > H (4)
- H is horizontal (front) → ❗reverse
- 1 → 2 → 3 = clockwise → reversed → S
✔️ Answer: (ii) → S
(iii)
Groups: NH₂, COOH, CH₃, H
Priority: NH₂ (1) > COOH (2) > CH₃ (3) > H (4)
- H is horizontal (front) → ❗reverse
- 1 → 2 → 3 = anticlockwise → reversed → R
✔️ Answer: (iii) → R
(iv) (Two chiral centers)
🔹 Upper carbon
Priority: NH₂ (1) > COOH (2) > lower C (3) > H (4)
- H is horizontal → reverse
- 1 → 2 → 3 = anticlockwise → reversed → R
🔹 Lower carbon
Priority: OH (1) > upper C (2) > CH₃ (3) > H (4)
- H is horizontal → reverse
- 1 → 2 → 3 = clockwise → reversed → S
✔️ Answer: (iv) → (R, S)
Final Answers:
- (i) → R
- (ii) → S
- (iii) → R
- (iv) → (R, S)
