1.Charge on Species (Most Important First)
Species with negative charge are stronger bases than neutral ones.
Example
Basicity is a measure of how well a compound (a base) shares its lone pair with a proton. The
stronger the base, the better it shares its electrons. Basicity is measured by an equilibrium constant
(the acid dissociation constant, Ka) that indicates the tendency of the conjugate acid of the base to
lose a proton
2. Electronegativity (Across a Period)
If atoms are in the same period, basicity decreases with electronegativity.
Less electronegative atom holds negative charge poorly → stronger base.
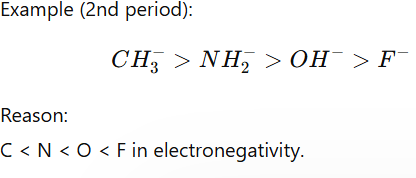
3.Hybridization Effect
More s-character stabilizes negative charge, making the base weaker.
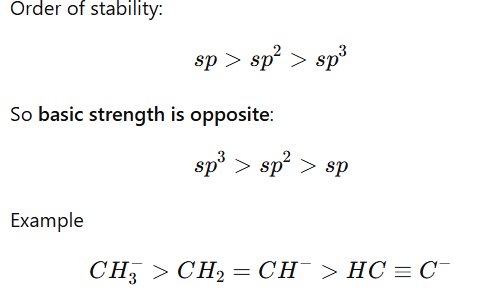
4.Resonance
If negative charge is delocalized, the base becomes less basic.
Example:
Acetate ion vs Ethoxide
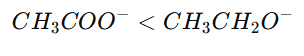
5.Inductive Effect
Electron-withdrawing groups (–I effect) decrease basicity.
Example
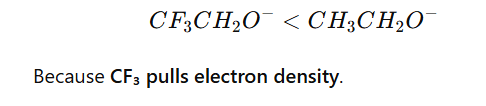
6. Aromaticity
If negative charge contributes to aromatic stabilization, the base becomes weaker.
Example
Cyclopentadienyl anion is stable → weak base.
Quick NEET/JEE Order (Decision Flow)
When comparing bases, check in this order:
1️⃣ Charge
2️⃣ Resonance
3️⃣ Electronegativity
4️⃣ Hybridization
5️⃣ Inductive effect
6️⃣ Aromaticity
7️⃣ Atom size
20 NEET / JEE MCQs – Basic Strength
1
Order of basicity:
CH₃⁻ , NH₂⁻ , OH⁻ , F⁻
A. CH₃⁻ > NH₂⁻ > OH⁻ > F⁻
B. NH₂⁻ > CH₃⁻ > OH⁻ > F⁻
C. OH⁻ > NH₂⁻ > CH₃⁻ > F⁻
D. F⁻ > OH⁻ > NH₂⁻ > CH₃⁻
2
Which is the strongest base?
A. NH₃
B. CH₃NH₂
C. (CH₃)₂NH
D. C₆H₅NH₂
3
Order of basicity:
NH₃ , CH₃NH₂ , (CH₃)₂NH , (CH₃)₃N (aqueous)
A. 2° > 1° > 3° > NH₃
B. 3° > 2° > 1° > NH₃
C. NH₃ > 1° > 2° > 3°
D. 1° > 2° > 3° > NH₃
4
Which is the weakest base?
A. Pyridine
B. Aniline
C. NH₃
D. CH₃NH₂
5
Correct order of basicity:
NH₂⁻ , OH⁻ , F⁻
A. NH₂⁻ > OH⁻ > F⁻
B. F⁻ > OH⁻ > NH₂⁻
C. OH⁻ > NH₂⁻ > F⁻
D. NH₂⁻ > F⁻ > OH⁻
6
Which is the strongest base?
A. HC≡C⁻
B. CH₂=CH⁻
C. CH₃⁻
D. C₆H₅⁻
7
Correct order of basicity:
CH₃NH₂ , C₆H₅NH₂ , NH₃
A. CH₃NH₂ > NH₃ > C₆H₅NH₂
B. NH₃ > CH₃NH₂ > C₆H₅NH₂
C. C₆H₅NH₂ > CH₃NH₂ > NH₃
D. NH₃ > C₆H₅NH₂ > CH₃NH₂
8
Which compound is most basic?
A. Pyrrole
B. Pyridine
C. Aniline
D. Ammonia
9
Which is strongest base?
A. CH₃O⁻
B. C₂H₅O⁻
C. OH⁻
D. F⁻
10
Order of basicity:
sp³ N , sp² N , sp N
A. sp³ > sp² > sp
B. sp > sp² > sp³
C. sp² > sp³ > sp
D. sp³ > sp > sp²
11
Which is least basic?
A. CH₃NH₂
B. NH₃
C. Aniline
D. (CH₃)₂NH
12
Which has maximum basicity?
A. NH₂⁻
B. OH⁻
C. H₂O
D. NH₃
13
Order of basicity:
Pyrrole , Pyridine , Piperidine
A. Piperidine > Pyridine > Pyrrole
B. Pyridine > Piperidine > Pyrrole
C. Pyrrole > Pyridine > Piperidine
D. Pyridine > Pyrrole > Piperidine
14
Which is stronger base?
A. CH₃NH₂
B. C₆H₅NH₂
15
Most basic compound:
A. NH₃
B. CH₃NH₂
C. C₂H₅NH₂
D. (CH₃)₂NH
16
Order of basicity:
NH₃ , PH₃ , AsH₃
A. NH₃ > PH₃ > AsH₃
B. AsH₃ > PH₃ > NH₃
C. PH₃ > NH₃ > AsH₃
D. NH₃ > AsH₃ > PH₃
17
Which is least basic?
A. Pyrrole
B. Pyridine
C. Piperidine
D. NH₃
18
Which is strongest base?
A. NH₂⁻
B. CH₃O⁻
C. OH⁻
D. NH₃
19
Order of basicity:
Aniline , p-methoxyaniline , p-nitroaniline
A. p-OCH₃ > aniline > p-NO₂
B. aniline > p-OCH₃ > p-NO₂
C. p-NO₂ > aniline > p-OCH₃
D. p-OCH₃ > p-NO₂ > aniline
20
Which is strongest base?
A. NH₃
B. CH₃NH₂
C. (CH₃)₂NH
D. (CH₃)₃N
Answer Key
- A
- C
- A
- B
- A
- C
- A
- B
- B
- A
- C
- A
- A
- A
- D
- A
- A
- A
- A
- C
