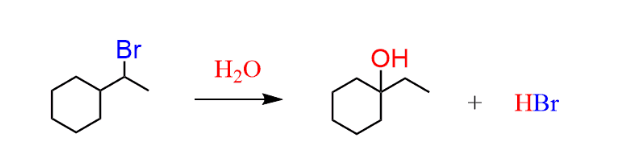
Ring Expansion (Carbocation Rearrangement)
Ring expansion is a type of carbocation rearrangement in which a smaller ring increases its size (usually by one carbon) to reduce ring strain.
It commonly occurs in SN1 or E1 reactions when a carbocation is adjacent to a small ring.
Why Ring Expansion Happens
Small rings such as:
- Cyclopropane
- Cyclobutane
have high angle strain.
When a carbocation forms next to these rings, a C–C bond shifts (1,2-shift) and the ring expands to:
- Cyclobutane
- Cyclopentane
which are more stable
PROBLEM :
Propose a mechanism for the following reaction:
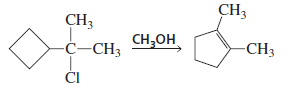
In this reaction the substrate is a tertiary alkyl chloride attached to a cyclobutane ring and the reagent is methanol (CH₃OH), which is a weak nucleophile and polar protic solvent.
So the reaction proceeds through an SN1 mechanism.
Step 1: Carbocation formation
The Cl⁻ leaves, forming a tertiary carbocation.
Step 2: Ring expansion (rearrangement)
Cyclobutane rings have high ring strain.
To reduce strain, a 1,2-shift of a ring bond occurs, converting:
cyclobutane → cyclopentane carbocation
This produces a more stable carbocation and also reduces ring strain.
Step 3: Deprotonation
Methanol removes a proton from the carbocation, giving an alkene.
Final Product
The rearranged product is:
1,2-dimethylcyclopentene
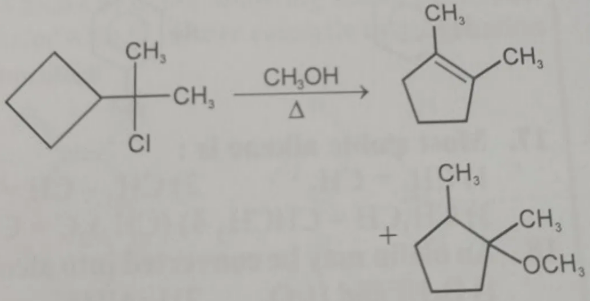
In this reaction substitution does NOT take place as the final product
Reason
Leaving group departure
Cl⁻ leaves first → tertiary carbocation forms (SN1 condition, because solvent is CH₃OH, weak nucleophile).
Carbocation rearrangement
The carbocation is next to a cyclobutane ring, which has high ring strain.
So a 1,2-C–C shift occurs, causing ring expansion:
Cyclobutane → Cyclopentyl carbocation
This rearrangement is very favorable because it relieves ring strain.
Elimination occurs
Instead of nucleophilic attack by methanol, a proton is removed, forming a stable alkene.
Why elimination dominates
- Rearranged carbocation becomes very stable
- Formation of cyclopentene double bond is thermodynamically favorable
- Relief of ring strain drives the reaction
Final product
1,2-dimethylcyclopentene
✔ Therefore substitution product (ether) is not observed as the major product.
The reaction proceeds via SN1 → rearrangement → E1 elimination.
Important teaching point (useful for students)
Whenever you see:
- Carbocation next to small ring (cyclobutane / cyclopropane)
- Ring expansion possible
➡ Rearranged alkene products are usually formed.
If you want, I can also show the step-by-step rearrangement mechanism diagram (very useful for explaining this in class).
Can cyclopentane undergo ring expansion?
Yes, but it is much less common.
Reason
Ring expansion mainly occurs to reduce ring strain.
Approximate ring strain order:
Cyclopropane > Cyclobutane > Cyclopentane > Cyclohexane
- Cyclopropane and cyclobutane have high strain → ring expansion very common
- Cyclopentane has very little strain → expansion is rare
So normally:
Cyclopentane → does NOT easily expand to cyclohexane
because there is no strong driving force.
When cyclopentane expansion can occur
It may happen when:
- A much more stable carbocation forms
- Substituted cyclohexane product becomes very stable
- Strong rearrangement conditions
Example idea
Cyclopentyl carbocation
→ 1,2-alkyl shift
→ Cyclohexyl carbocation
But this is far less favorable than:
Cyclobutane → Cyclopentane expansion.
Important Exam Rule (JEE / NEET)
Students should remember:
| Ring | Ring Expansion |
|---|---|
| Cyclopropane | Very common |
| Cyclobutane | Common |
| Cyclopentane | Rare |
| Cyclohexane | Almost never |
Simple memory trick
Small rings expand. Stable rings do not.
So usually:
3 → 4 → 5 expansion happens easily
but 5 → 6 rarely occur
PROBLEM