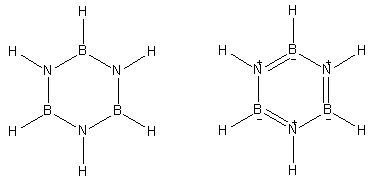
Structure Overview
- Ring: B–N–B–N–B–N (6-membered ring)
- Similar to Benzene
- Planar structure
Hybridisation of Atoms
1. Boron (B)
- Hybridisation = sp²
- Forms:
- 2 σ bonds with N
- 1 σ bond with H
- Has empty p-orbital
2. Nitrogen (N)
- Hybridisation = sp²
- Forms:
- 2 σ bonds with B
- 1 σ bond with H
- Has lone pair in p-orbital (participates in π bonding)
π-Bonding (Important Concept)
- p-orbitals of B and N overlap → π system
- But unlike benzene:
- π electrons are not equally distributed
- Due to electronegativity difference (B < N)
So, borazine is:
- Partially aromatic
- Less stable than benzene
Key Differences from Benzene
| Property | Borazine | Benzene |
|---|---|---|
| Hybridisation | All atoms sp² | All C sp² |
| Bond nature | Polar (B–N) | Nonpolar |
| Aromaticity | Weak | Strong |
Exam Points (JEE/NEET)
✔ Both B and N are sp² hybridised
✔ Structure is planar hexagon
✔ Contains delocalised π electrons
✔ Called “inorganic benzene” due to similarity
