Sublimation
What is Sublimation?
Sublimation is the process in which a solid directly changes into vapour without becoming liquid.
Example:
- Ammonium chloride
- Iodine
- Camphor
- Naphthalene
Why does it happen?
Some solids have:
- Weak intermolecular forces
- High vapour pressure even at lower temperatures
So, when heated, they directly escape into vapour form.
Sublimation as a Separation Technique
It is used to purify substances:
✔ Sublimable substance → turns into vapour
✔ Non-sublimable impurities → remain as solid
Then vapour is cooled → forms pure solid again.
Simple Setup
- Heat the mixture
- Sublimable substance vaporizes
- Vapour condenses on a cold surface
- Pure solid is collected
Example Use
Separating:
- Ammonium chloride + sand
Ammonium chloride sublimates, sand stays behind.
Crystallisation
What is Crystallisation?
A purification technique used to purify solid organic compounds based on difference in solubility.
Principle
- Substance is less soluble at low temperature
- Substance is more soluble at high temperature
So:
- Dissolve at high temp
- Cool → pure crystals form
Steps
- Dissolve impure solid in hot solvent
- Make a saturated solution
- Filter (if needed, remove insoluble impurities)
- Cool the solution
- Pure crystals separate out
- Filter and dry crystals
Important Terms
- Mother liquor → remaining liquid after crystallisation
- Activated charcoal → removes colored impurities
🔹 Examples
Example 1: Benzoic Acid purification
- Impure benzoic acid dissolved in hot water
- On cooling → pure crystals of benzoic acid form
When repeated crystallisation is needed?
When
Simple Distillation (large boiling point difference)
When used:
- Difference in b.p. > 25–30°C
- Or liquid + non-volatile impurity
Examples:
Example 1:
- Water + salt
Water distils, salt remains
Example 2:
- Chloroform (334 K) + Aniline (457 K)
Easily separated
Example 3:
- Alcohol + sugar solution
Alcohol vaporises, sugar stays
Example 4:
- Acetone + water
Acetone (low b.p.) comes first
Fractional Distillation (close boiling points)
When used:
- Difference in b.p. < 25°C
Examples:
Example 1:
- Ethanol (78°C) + Water (100°C)
Cannot separate by simple distillation
Example 2:
- Benzene (80°C) + Toluene (110°C)
Need fractionating column
Example 3:
- Hexane + Heptane
Very close boiling points
Example 4 (Industrial):
- Crude oil refining
Petrol, diesel, kerosene separated
3. Steam Distillation (heat-sensitive substances)
When used:
- Substance:
- Immiscible with water
- Volatile with steam
- Decomposes at high temperature
Examples:
Example 1:
- Extraction of essential oils
From plants (e.g., clove oil, eucalyptus oil)
Example 2:
- Aniline purification
High b.p. but steam volatile
Example 3:
- Nitrobenzene
Separated using steam
Example 4:
- Rose oil extraction (attar)
Used in perfumes
Quick Comparison (Very Important)
| Method | Condition | Example |
|---|---|---|
| Simple Distillation | Large b.p. difference | Water + salt |
| Fractional Distillation | Small b.p. difference | Ethanol + water |
| Steam Distillation | Heat-sensitive, steam volatile | Essential oils |
Distillation Under Reduced Pressure (Vacuum Distillation)
Statement:
Distillation under reduced pressure → Glycerol + spent-lye
Why this method is used?
Some liquids:
- Have very high boiling points
- Decompose before boiling
Example: Glycerol
Principle
When pressure is reduced:
➡ Boiling point decreases
So, substance can distil at lower temperature without decomposition
Application in given case
Mixture:
- Glycerol (high b.p., heat sensitive)
- Spent-lye (impurities)
What happens?
- Pressure ↓
- Glycerol boils at lower temp
- Vapours collected → pure glycerol
- Impurities remain
Why not simple distillation?
❌ Glycerol decomposes at high temperature
✔ So vacuum distillation is required
🔹 More Examples
Example 1:
- Glycerol purification (most common)
Example 2:
- High boiling oils
- Example 3:
- Fatty acids
Example 4:
- Petroleum residues
Quick Summary
| Method | Used for |
|---|---|
| Simple distillation | Low b.p. liquids |
| Fractional distillation | Close b.p. liquids |
| Steam distillation | Heat-sensitive, steam volatile |
| Reduced pressure distillation | High b.p., decomposing liquids |
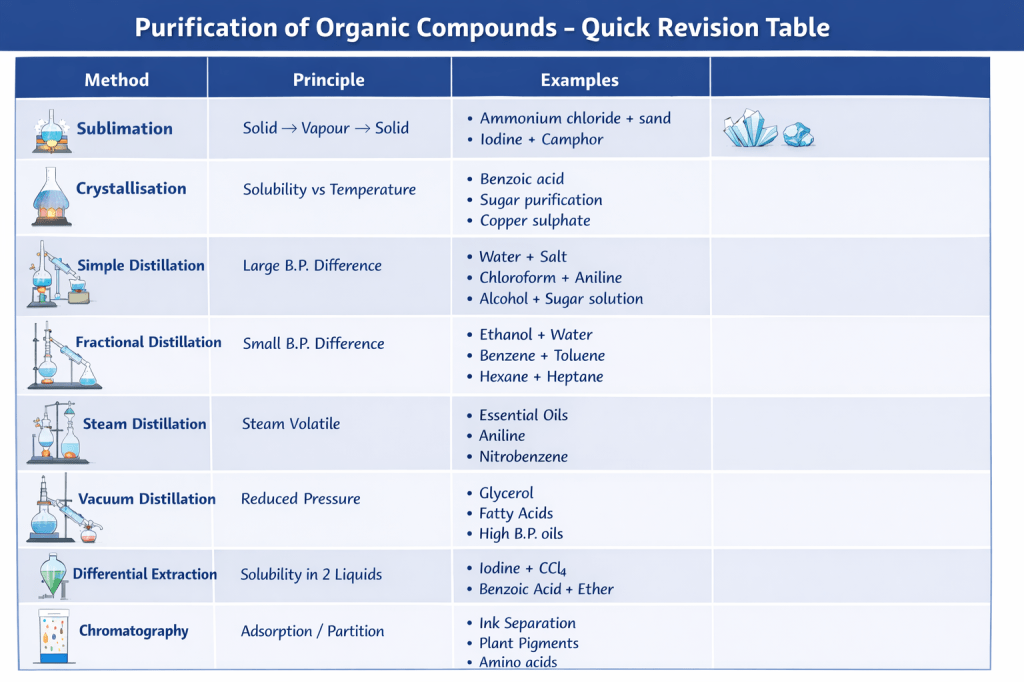
Differential Extraction
When an organic compound is present in an aqueous medium, it is separated by shaking it with an organic solvent in which it is more soluble than in water. The organic solvent and aqueous solution should be immiscible so that they form two distinct layers which can be separated using a separating funnel. The organic solvent is later removed by distillation or evaporation to obtain the compound.
Concept: Differential (Liquid–Liquid) Extraction
Principle
Based on difference in solubility of a compound in two immiscible liquids
Usually:
- Water (aqueous layer)
- Organic solvent (like ether, benzene)
Key Conditions
✔ Two liquids must be immiscible
✔ Compound should be more soluble in organic solvent
Process (Separating Funnel)
- Take mixture in separating funnel
- Add organic solvent
- Shake well
- Allow layers to separate
Two layers form:
- Upper layer (usually organic solvent)
- Lower layer (water)
- Separate layers
- Evaporate solvent → get pure compound
Example
Example 1:
- Iodine in water + carbon tetrachloride (CCl₄)
→ Iodine moves to organic layer
Example 2:
- Benzoic acid from water using ether
Continuous Extraction
When used?
- When compound is slightly soluble in solvent
Why?
- Single extraction inefficient
- Repeated extraction increases yield
Same solvent reused again and again
Advantages
- Efficient separation
- Better recovery of compound
- Widely used in organic chemistry labs
Chromatography
What is Chromatography?
A separation technique where components of a mixture are separated based on their different affinities between two phases.
Principle
Based on distribution of components between two phases:
- Stationary phase → fixed (solid or liquid)
- Mobile phase → moving (liquid or gas)
Different substances move at different speeds → separation occurs
Types of Chromatography
1. Paper Chromatography
- Stationary phase → paper
- Mobile phase → solvent
Used for:
- Ink separation
- Plant pigments
2. Thin Layer Chromatography (TLC)
- Stationary phase → silica gel layer
- Faster and more accurate than paper chromatography
3. Column Chromatography
- Column filled with adsorbent (silica/alumina)
- Used for larger scale separation
4. Gas Chromatography (GC)
- Mobile phase → gas
- Used for volatile compounds
How Separation Happens?
- Component with more attraction to stationary phase → moves slowly
- Component with more attraction to mobile phase → moves faster
Example
Ink Separation
Black ink → separates into different colors on paper
Plant Pigments
Chlorophyll, carotene separated using chromatography
Important Term: Rf Value
Uses
✔ Separation of mixtures
✔ Purification
✔ Checking purity
✔ Drug testing
✔ Food analysis
