According to Bohr, electrons move in stationary orbits where they do not radiate energy, hence energy of an orbit remains constant with time.
Bohr assumed stationary orbits (not proved mathematically) This explains atomic stability Energy is quantized and constant in each orbit.
Electrostatic force = Centripetal force
Step 1: Electron is attracted to nucleus, so:
Step 2 : Quantization of angular momentum
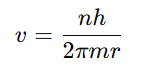
Step 3: Substitute v into Step 1
Step 4: Simplify
Step 5: Solve for radius r
Simplify:
Final Result
where,
Key Results
-
- Valid for hydrogen-like species (H, He⁺, Li²⁺)
Bohr model, hydrogen-like atom, radius derivation, angular momentum quantization, electrostatic force
