Primary and secondary alkyl halides can undergo SN2 and E2 reactions.
Tertiary alkyl halides generally undergo E2 reactions or SN1 and E1 reactions.
Tertiary alkyl halides do not undergo SN2 reactions because of steric hindrance.
If the reagent is a good nucleophile and a strong base, a tertiary alkyl halide undergoes an E2 reaction.
If the weakly basic solvent is the only nucleophile/base, the tertiary alkyl halide undergoes SN1 and E1 reactions.
Because a primary alkyl halide is the most reactive in an SN2 reaction, substitution will
win the competition.
Case 1 : SN2/E2 Reactions of Primary Alkyl Halides
Problem: Bromopropane + CH₃O⁻ (methoxide ion)
First identify the substrate. Bromopropane (1-bromopropane) is a primary alkyl halide, and CH₃O⁻ is a strong nucleophile and strong base.
Because the substrate is primary, SN2 reaction dominates over E2.
Reaction
CH₃CH₂CH₂Br + CH₃O⁻ → CH₃CH₂CH₂OCH₃ + Br⁻
Major Product
Methoxypropane (methyl propyl ether)
Structure:
CH₃CH₂CH₂OCH₃ (90%)
Minor Product (possible but small amount)
E2 elimination → Propene (CH₂=CHCH₃) (10%)
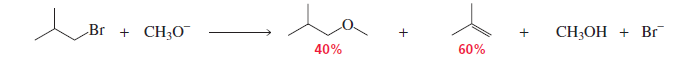
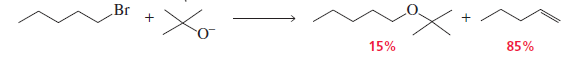
Steric Hindrance Favors the Elimination Product (E2.)
Case II : SN2/E2 Reactions of Primary Alkyl Halides
The stronger and bulkier the nucleophile/base, the greater the percentage of the elimination product.
The weaker and less bulky the nucleophile/base, the greater the percentage of the substitution product
eaction:
2-Chloropropane + Acetate ion (CH₃COO⁻)
Substrate: Secondary alkyl halide
Reagent: Acetate ion → good nucleophile but weak base
Reaction Type
Because acetate ion is a good nucleophile and weak base, the reaction mainly proceeds through substitution (SN2) rather than elimination.
Reaction
(CH₃)₂CHCl + CH₃COO⁻ → (CH₃)₂CH–OCOCH₃ + Cl⁻
Major Product
Isopropyl acetate (propan-2-yl acetate)
Structure:
CH₃–CH(OCOCH₃)–CH₃
Reason
- Secondary alkyl halide
- Weak base → elimination not favored
- Good nucleophile → substitution product dominates
Major product: Isopropyl acetate (100%)
2-Chloropropane + Ethoxide ion (C₂H₅O⁻)
Substrate
- 2-Chloropropane → secondary alkyl halide
Reagent
- Ethoxide ion (C₂H₅O⁻) → strong base and good nucleophile
Reaction Type
With a secondary alkyl halide and a strong base, E2 elimination is the major reaction.
Reaction
(CH₃)₂CHCl + C₂H₅O⁻ → CH₃–CH=CH₂ + C₂H₅OH + Cl⁻
Major Product
Propene
Structure:
CH₃–CH=CH₂
Reason
- Secondary substrate
- Strong base present
- Elimination favored over substitution
Minor Product (possible)
Isopropyl ethyl ether via SN2.
Major product: Propene (E2 elimination) (75%)
Minor Product
Isopropyl ethyl ether
(IUPAC: 2-ethoxypropane) (25%)
A Bulky Base Favors the Elimination Product
Bulky bases such as DBN and DBU are strong bases but poor nucleophiles because their bulky structure makes it difficult to attack the carbon atom.
When a secondary (2°) alkyl halide reacts with these bulky bases:
- SN2 substitution is hindered due to steric bulk around the base.
- The base instead abstracts a β-hydrogen from the alkyl halide.
- This leads to E2 elimination rather than substitution.
- Therefore, alkene formation becomes the major product.
- In many cases, bulky bases may favor the Hofmann (less substituted) alkene.
Conclusion:
With 2° alkyl halides, bulky bases like DBN and DBU mainly give E2 elimination products (alkenes) rather than substitution products.
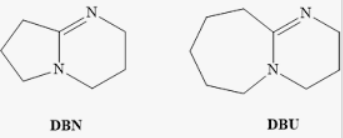
The full forms are:
- DBN
DBN = 1,5-Diazabicyclo[4.3.0]non-5-ene - DBU
DBU = 1,8-Diazabicyclo[5.4.0]undec-7-ene
A High Temperature Favors the Elimination Product
2-Bromopropane + OH⁻
Substrate: Secondary alkyl halide
Both substitution (SN2) and elimination (E2) can occur.
Temperature decides the major product.
At 45 °C (lower temperature)
Lower temperature favors substitution.
Reaction (SN2):
(CH₃)₂CHBr + OH⁻ → (CH₃)₂CHOH + Br⁻
Minor product:
2-Propanol (Isopropyl alcohol) (47%)
Major product: Propene (53%)
At 100 °C (higher temperature)
Higher temperature favors elimination.
Reaction (E2):
(CH₃)₂CHBr + OH⁻ → CH₃–CH=CH₂ + H₂O + Br⁻
Major product:
Propene (71%)
Minor product: 2-Propanol (29%)
CASE III: E2 Reaction of a Tertiary Alkyl Halide
Because a tertiary alkyl halide cannot undergo an SN2 reaction, only an elimination product is
formed when a tertiary alkyl halide reacts with a strong base.
Tert-butyl bromide + Ethoxide ion (C₂H₅O⁻)
Substrate: Tertiary alkyl halide
Reagent: Strong base and good nucleophile
Key Concept
- Tertiary alkyl halides do not undergo SN2 reactions due to strong steric hindrance.
- When a strong base like ethoxide ion is present, the reaction proceeds mainly by E2 elimination.
Reaction (E2)
Major Product
2-Methylpropene (Isobutene)
Structure:
(CH₃)₂C=CH₂
Reason
- Substrate is tertiary
- SN2 is blocked due to steric hindrance
- Strong base present
- Therefore E2 elimination dominates
Major product: 2-Methylpropene (alkene) (100%)
CASE IV: SN1/E1 Reactions of Tertiary Alkyl Halides
Tert-butyl bromide + Ethanol
Substrate: tertiary alkyl halide
Reagent/Solvent: ethanol (weak nucleophile and weak base, polar protic solvent)
Reaction Type
Tertiary alkyl halides in a weak nucleophile solvent undergo SN1 and E1 reactions.
Step 1: Formation of tert-butyl carbocation
Step 2: Nucleophile or base reacts with the carbocation.
Substitution Reaction (SN1) – Major Product
Major product:
tert-Butyl ethyl ether (81%)
Structure:
Elimination Reaction (E1) – Minor Product
Minor product:
2-Methylpropene (isobutene) (19 %)
tert-Butyl bromide + Ethoxide ion (C₂H₅O⁻)
Substrate: tertiary alkyl halide
Reagent: strong base (ethoxide ion)
Reaction Type
- SN2 cannot occur because the tertiary carbon is highly sterically hindered.
- With a strong base, the reaction proceeds mainly by E2 elimination.
Reaction (E2)
Major Product
2-Methylpropene (Isobutene) (100%)
Structure:
(CH₃)₂C=CH₂
Reason
- Substrate is tertiary.
- Strong base present.
- SN2 blocked due to steric hindrance.
- Therefore E2 elimination dominates.
Major product: 2-Methylpropene (alkene).

