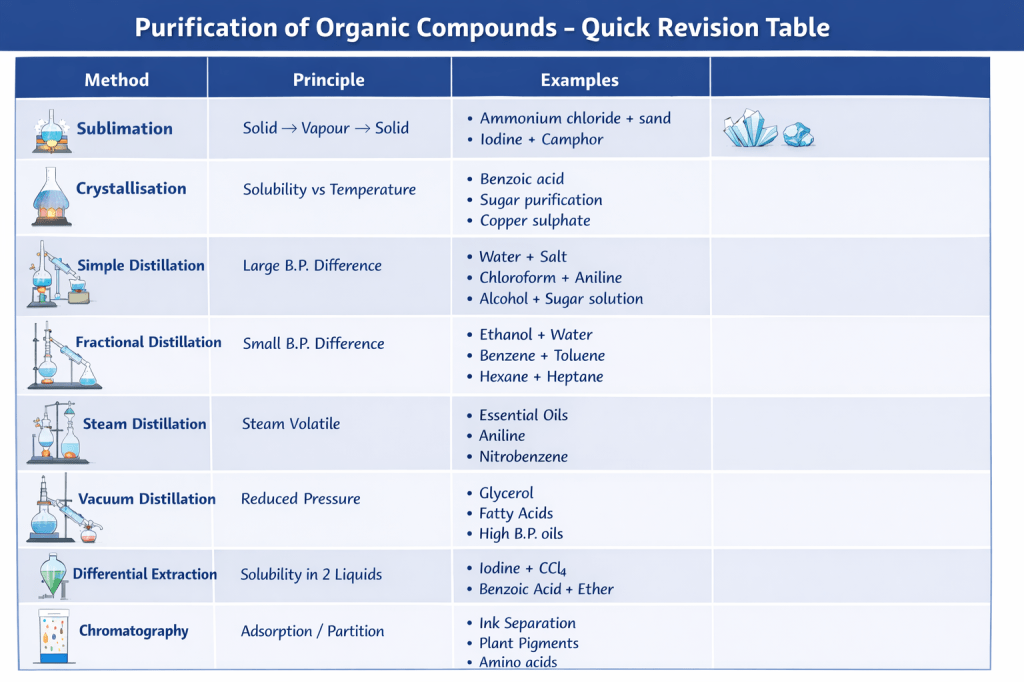
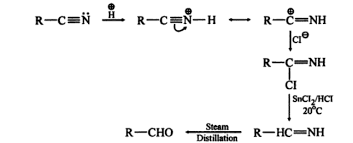
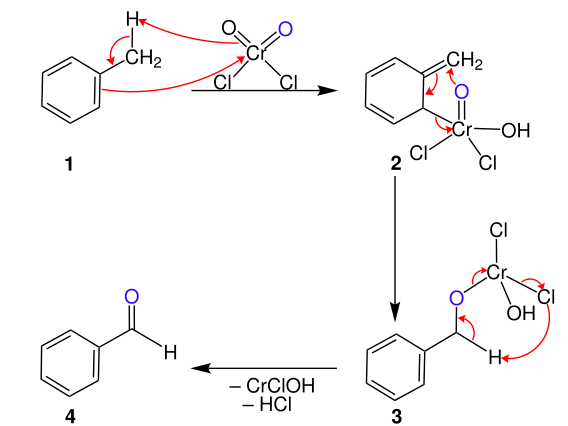
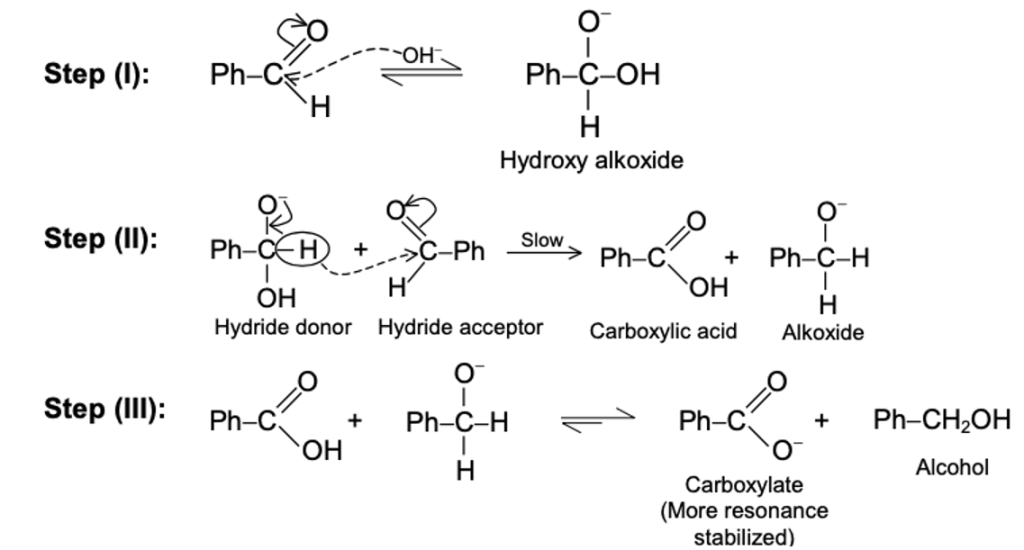
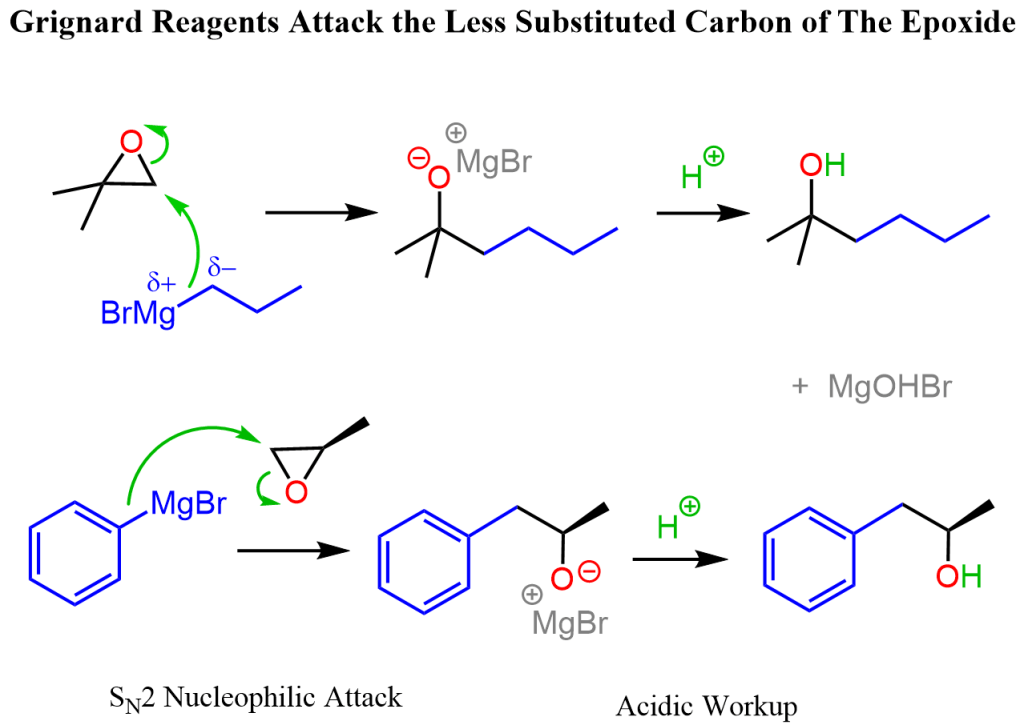
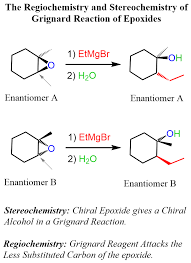
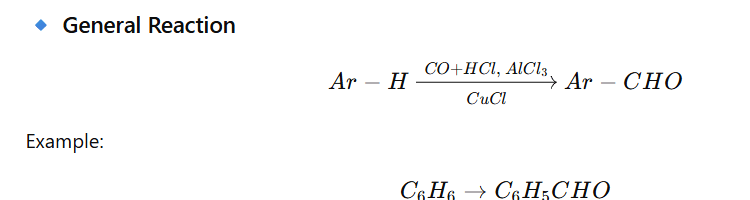SECTION 1: SOLUTIONS (10 MCQs)
- Which of the following is an ideal solution?
A) Acetone + Chloroform
B) Benzene + Toluene
C) Ethanol + Water
D) Water + HCl - Raoult’s law is applicable to:
A) Non-volatile solutes
B) Electrolytes
C) Ideal solutions
D) Strong acids - Mass of glucose required to be dissolved to prepare one litre of its solution which is isotonic with 15 g solution of urea is(Given: Molar mass in g C : 12, H : 1, O : 16, N : 14)
A) 55g
B) 30g
C) 15g
D) 45g - The Henry’s law constant (KH) values of three gases (A, B, C) in water are 145, 2 × 10-5 and 35 kbar, respectively. The solubility of these gases in water follow the order:
A) B > A > C (B) B > C > A (C) A > C > B (D) A > B > C - The plot of osmotic pressure ( ∏ ) vs concentration ( mol− 1 ) for a solution gives a straight line with slope 25.73 L bar mol−1 . The temperature at which the osmotic pressure measurement is done is (Use R = 0.083 L bar mol−1 K −1 )
A) 1
B) 2
C) 3
D) 0 - Osmotic pressure is given by:
A) Boyle’s law
B) Ideal gas equation
C) van’t Hoff equation
D) Dalton’s law
- Maximum boiling azeotrope shows:
A) Positive deviation
B) Negative deviation
C) No deviation
D) Infinite deviation - Which increases boiling point?
A) Adding volatile solute
B) Adding non-volatile solute
C) Decreasing pressure
D) Increasing solvent - Henry’s law relates:
A) Pressure and solubility
B) Volume and temperature
C) Mass and density
D) Temperature and pressure - Which amongst the following aqueous solution of electrolytes will have minimum elevation in boiling point? Choose the correct option :-
A) 0.05 M NaCl
B) 0.1 M KCl
C) 0.1MgSO4
D) 1M NaCl
SECTION 2: CLASSIFICATION OF ELEMENTS (10 MCQs)
- Which of the following statements are true?
A) Unlike Ga that has a very high melting point, Cs has a very low melting point.
B) On Pauling scale, the electronegativity values of N and Cl are not the same
C) A r , K + , C l − , C a 2 + , and S 2 − are all isoelectronic species.
D) The correct order of the first ionization enthalpies of N a , M g , A l and Si is S i > A l > M g > Na

12. Which among the following electronic configurations belong to main group elements?
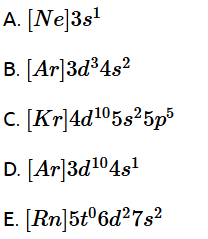
Choose the correct answer from the option given below :
(1) B and E only
(2) A and C only
(3) D and E only
(4) A, C and D
13.The correct decreasing order of atomic radii (pm) of Li, Be, B and C is
(1) Be > Li > B > C
(2) Li > Be > B > C
(3) C > B > Be > Li
(4) Li > C > Be > B
14.Match List-I with List-II :

Identify the correct answer from the options given below :
(1) A-III, B-I, C-IV, D-II
(2) A-I, B-IV, C-III, D-II
(3) A-II, B-IV, C-I, D-III
(4) A-III, B-IV, C-I, D-II
15.Arrange the following elements in increasing order of first ionization enthalpy: Li, Be, B, C, N
Choose the correct answer from the options given below:
(1) Li < Be < B < C < N
(2) Li < B < Be < C < N
(3) Li < Be < C < B < N
(4) Li < Be < N < B < CR
16. Arrange the following elements in increasing order of electronegativity:
N, O, F, C, Si
Choose the correct answer from the options given below :
Choose the correct answer from the options given below:
(1) Si < C < N < O < F
(2) Si < C < O < N < F
(3) O < F < N < C < Si
(4) F < O < N < C < Si
17 Which of the following is correctly matched?
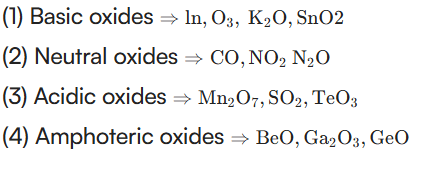
18. The correct sequence given below containing neutral, acidic, basic and amphoteric oxide each, respectively, is
Options: A. NO, ZnO, CO₂, CaO
B. ZnO, NO, CaO, CO₂
C. NO, CO₂, ZnO, CaO
D. NO, CO₂, CaO, ZnO
19. For elements B, C, N, Li, Be, O and F the correct order of first ionization enthalpy is
(1) Li < Be < B < C < N < O < F
(2) B > Li > Be > C > N > O > F
(3) Li < B < Be < C < O < N < F
(4) Li < Be < B < C < O < N < F
20. Decrease in size from left to right in actinoid series is greater and gradual than that in lanthanoid series due to
1.4f orbitals are second to last
2. 4f orbitals have greater shielding effect
3. 5f orbitals have poor shielding effect
4.5f orbitals have greater shielding effect
SECTION 3: d-BLOCK ELEMENTS (10 MCQs)
- Transition elements have:
A) Completely filled d-orbitals
B) Partially filled d-orbitals
C) No d-orbitals
D) Only s-orbitals - General configuration:
A) (n-1)d¹–¹⁰ ns¹–²
B) ns² np⁶
C) ns¹
D) np⁵ - Highest oxidation state of Mn:
A) +2
B) +4
C) +7
D) +6 - Color in transition metals due to:
A) s-electrons
B) d-d transition
C) p-electrons
D) nuclear charge - Which is not transition element?
A) Zn
B) Fe
C) Cu
D) Cr - Catalytic property due to:
A) Low density
B) Variable oxidation states
C) High melting point
D) Atomic size - Magnetic behavior depends on:
A) paired electrons
B) unpaired electrons
C) protons
D) neutrons - Which is diamagnetic?
A) Fe²⁺
B) Mn²⁺
C) Zn²⁺
D) Cu²⁺ - Alloy of Cu and Zn:
A) Bronze
B) Brass
C) Steel
D) Solder - Highest melting point:
A) Hg
B) Fe
C) W
D) Cu
SECTION 4: COORDINATION COMPOUNDS (15 MCQs)
- Coordination number of central atom is:
A) number of ligands
B) number of bonds
C) number of donor atoms
D) oxidation state - Ligand donating one pair:
A) Bidentate
B) Monodentate
C) Tridentate
D) Polydentate - Example of bidentate ligand:
A) NH₃
B) H₂O
C) en
D) Cl⁻ - IUPAC name of [Cu(NH₃)₄]²⁺:
A) Copper tetraammine
B) Tetraammine copper(II)
C) Ammine copper
D) Copper ammonia - Oxidation state of Fe in [Fe(CN)₆]³⁻:
A) +2
B) +3
C) +6
D) −3 - Coordination compounds show isomerism:
A) Yes
B) No
C) Rarely
D) Only optical - Geometrical isomerism in:
A) tetrahedral
B) square planar
C) linear
D) trigonal - Strong field ligand:
A) Cl⁻
B) H₂O
C) CN⁻
D) Br⁻ - Crystal field splitting occurs in:
A) s-orbitals
B) p-orbitals
C) d-orbitals
D) f-orbitals - High spin complex has:
A) pairing
B) maximum unpaired electrons
C) no electrons
D) stable structure - Hybridization of [Ni(CN)₄]²⁻:
A) sp³
B) dsp²
C) sp²
D) d²sp³ - Werner theory explains:
A) valency
B) bonding
C) structure of complexes
D) reactivity - Chelate effect increases:
A) instability
B) stability
C) reactivity
D) acidity - [Co(NH₃)₆]³⁺ is:
A) paramagnetic
B) diamagnetic
C) ferromagnetic
D) antiferromagnetic - Which shows optical isomerism?
A) [Co(en)₃]³⁺
B) [NiCl₄]²⁻
C) [PtCl₄]²⁻
D) [Zn(NH₃)₄]²⁺