- Picric acid has three –NO₂ groups which are strong electron-withdrawing groups.
- These groups stabilize the conjugate base (picrate ion) by resonance and –I effect.
- Formic acid has only one –COOH group, so its conjugate base is less stabilized.
- Greater stabilization of conjugate base ⇒ higher acidity, so picric acid is stronger.
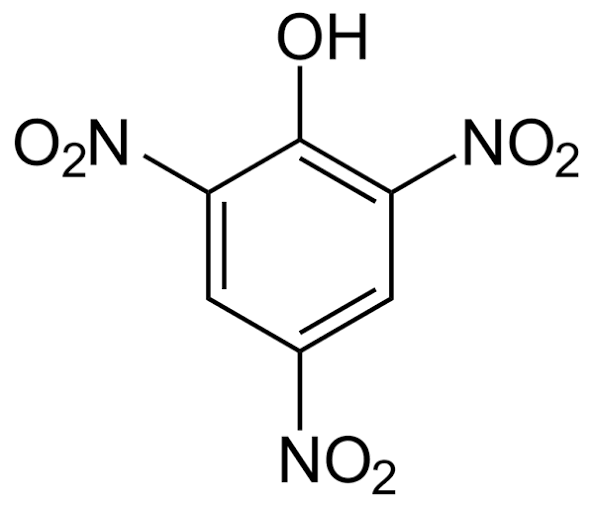
HCOOH (Picric Acid)
Picric acid is soluble in NaHCO₃ solution?
Picric acid (2,4,6-trinitrophenol) is a strong acid due to the presence of three –NO₂ groups, which strongly withdraw electrons and stabilize the phenoxide ion. Because of this high acidity, it can react even with a weak base like NaHCO₃.
Reaction:
Picric acid + NaHCO₃ → Sodium picrate (soluble) + CO₂ + H₂O
So, it dissolves in NaHCO₃ with effervescence of CO₂ gas.
