tephen Reaction (Stephen Aldehyde Synthesis) – Mechanism (JEE/NEET Concept)
Definition:
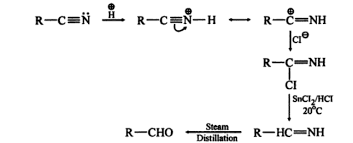
Conversion of nitriles (R–C≡N) into aldehydes (R–CHO) using SnCl₂/HCl, followed by hydrolysis.
Overall Reaction
Stepwise Mechanism
Step 1: Formation of Iminium Salt (Reduction Step)
- SnCl₂ (mild reducing agent) donates electrons.
- Nitrile gets partially reduced.
- Formation of iminium chloride salt:
Key point:
- Triple bond (C≡N) → double bond (C=NH)
- Controlled reduction (not all the way to amine)
Step 2: Hydrolysis of Iminium Salt
- Iminium salt reacts with water.
- Produces aldehyde + ammonium salt
R−CH=NH⋅HCl+H2O→R−CHO+NH4Cl
Important Points (Exam-Oriented)
- Works best for alkyl nitriles (aryl nitriles give poor yield).
- Stops at aldehyde stage (unlike strong reduction → amine).
- SnCl₂/HCl = selective mild reducing system
- Intermediate = iminium salt (not imine directly)
Shortcut Trick (Memory Tip)
“Nitrile + Mild Reduction = Aldehyde”
Think: Stephen = Stops Early (Aldehyde, not amine)
Example
