What is it?
Introduction of a formyl group (–CHO) into an aromatic ring using:
CO + HCl in presence of Aluminium chloride and Cuprous chloride
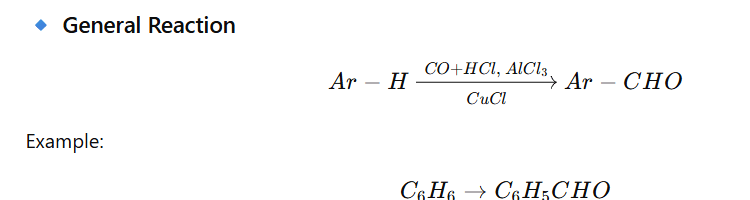
Mechanism (Stepwise)
Formation of Electrophile
- CO + HCl react in presence of AlCl₃/CuCl
- Generate formyl cation
Electrophilic Attack (EAS Step)
- Benzene ring attacks HCO⁺
- Forms sigma complex (arenium ion)
Deprotonation
- Loss of H⁺ restores aromaticity
- Forms benzaldehyde complex
Hydrolysis
- Final hydrolysis gives aldehyde (–CHO)
Key Points for Exams
✔ Type of Reaction
Electrophilic Aromatic Substitution (EAS)
✔ Important Conditions
- CO must be dry
- Presence of both AlCl₃ and CuCl is essential
❌ Limitations
- Does NOT work with strongly deactivated rings
(e.g., nitrobenzene ❌)
🔁 Comparison Trick
| Reaction | Reagent | Product |
|---|---|---|
| Gattermann–Koch | CO + HCl | –CHO |
| Friedel–Crafts | R–Cl / RCOCl | –R / –COR |
🔹 One-Line Revision
Gattermann–Koch introduces –CHO on benzene via formyl cation (HCO⁺) using CO + HCl / AlCl₃–CuCl
