Oxidation of a benzylic methyl group (–CH₃ attached to benzene) to an aldehyde (–CHO) using
Chromyl chloride in a non-aqueous solvent (like CCl₄).
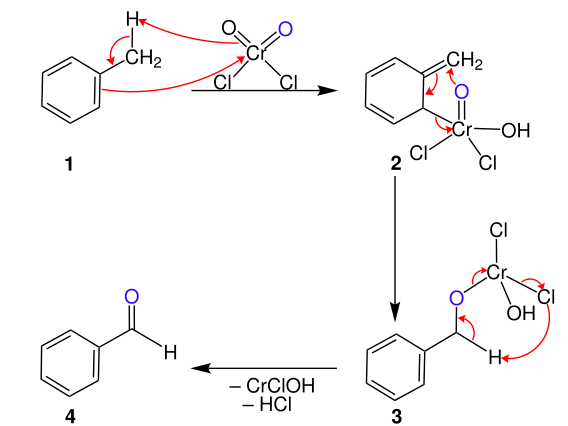
Mechanism (Stepwise)
Formation of Etard Complex
- Chromyl chloride attacks benzylic hydrogen
- Forms a brown complex (Etard complex)
Hydrolysis of Complex
- On hydrolysis (H₂O), the complex breaks
- Gives aldehyde
