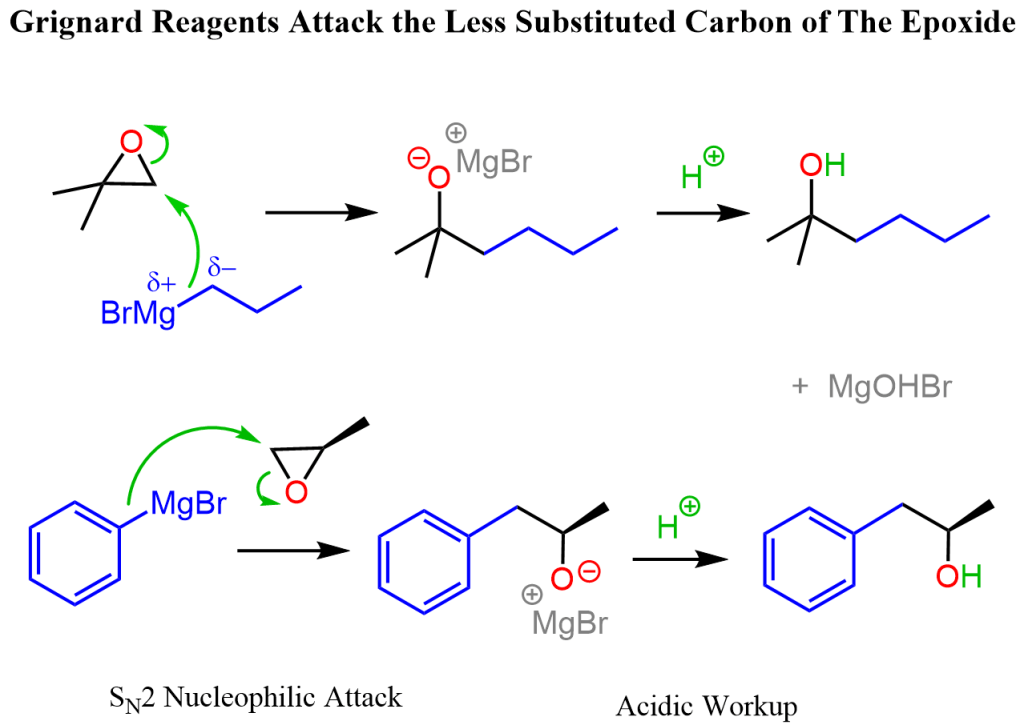
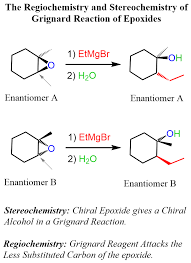
This is a high-weightage concept from alcohols + organometallics.
General Reaction:
Core Mechanism:
- Grignard reagent (RMgX) behaves like R⁻ (strong nucleophile)
- Attacks epoxide → ring opening (SN2 type)
- Final step: acidic hydrolysis → alcohol
Golden Rules (VERY IMPORTANT):
Attack Position:
✔ Always attacks LESS substituted carbon (SN2 mechanism)
Product Type:
✔ Alcohol is formed at the carbon where O⁻ was present
✔ Nature depends on epoxide structure:
- Ethylene oxide → Primary alcohol
- Substituted epoxide → Secondary / tertiary alcohol
Chain Extension Trick:
✔ Adds +2 carbons (only with ethylene oxide clearly)
✔ With other epoxides → chain increases but depends on structure
Important Cases:
1. Ethylene Oxide:
Always primary alcohol
+2 carbon extension (fixed)
2. Unsymmetrical Epoxide (e.g., Propylene oxide):
Attack at less hindered carbon
Usually secondary alcohol
- JEE/NEET Quick Summary:
✔ RMgX = nucleophile (R⁻)
✔ Epoxide opening = SN2 reaction
✔ Attack = less substituted carbon
✔ Product = alcohol after H₃O⁺