Cannizzaro Reaction (JEE/NEET Concepts)
Definition:
Cannizzaro reaction is a disproportionation reaction in which an aldehyde without α-hydrogen undergoes self oxidation–reduction in the presence of strong base (NaOH/KOH).
General Reaction
(One molecule is reduced → alcohol, other oxidized → carboxylate salt)
- Key Conditions (VERY IMPORTANT for JEE)
- Aldehyde must have NO α-H (alpha hydrogen)
- Strong base: conc. NaOH or KOH
- Usually occurs with:
- Formaldehyde (HCHO)
- Benzaldehyde (C₆H₅CHO)
Example
(Benzaldehyde → benzyl alcohol + sodium benzoate)
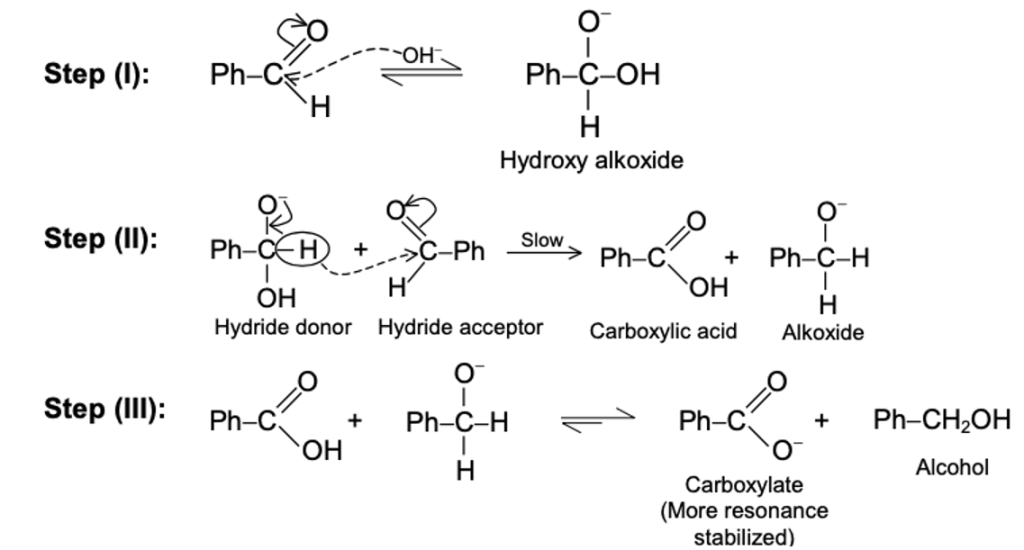
Mechanism (Conceptual Steps)
- Nucleophilic attack of OH⁻ on aldehyde → alkoxide intermediate
- Hydride transfer (H⁻ shift) from one molecule to another
- Formation of:
- Alcohol (reduction)
- Carboxylate ion (oxidation)
Types of Cannizzaro Reaction
- Self Cannizzaro
Same aldehyde reacts
Example: benzaldehyde - Cross Cannizzaro (Important!)
Two different aldehydes- One should be formaldehyde (HCHO) (best reducing agent)
(Formaldehyde gets oxidized, other gets reduced)
