The Kolbe’s reaction (Kolbe–Schmitt reaction) is used to introduce a –COOH group into phenol, mainly at the ortho position.
Overall reaction
Phenol + NaOH + CO₂ (pressure, heat) → o-hydroxybenzoic acid (salicylic acid)
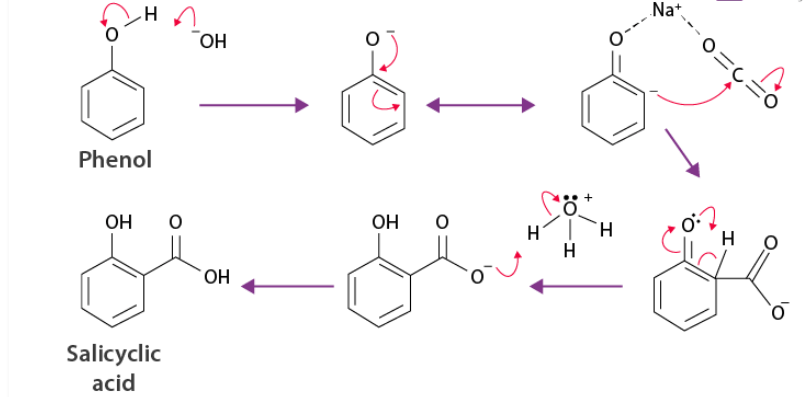
Key Points
- Reagents: NaOH + CO₂ (high pressure, ~125°C)
- Electrophile: CO₂
- Major product: ortho (salicylic acid)
- Para product is minor
- Ortho favored due to intramolecular H-bonding
