Leaving Group Ability (Best → Worst)
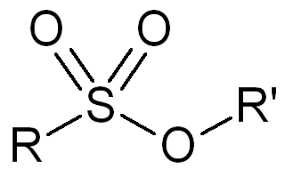
Sulfonate ester

Sulfonium ion structure
Why HO⁻ and RO⁻ are Poor Leaving Groups
- HO⁻ (hydroxide ion) and RO⁻ (alkoxide ion) are:
- Strong bases
- High-energy species
- A good leaving group should be:
- Stable after leaving
- Weak base
Since HO⁻ and RO⁻ are strong bases, they are unstable when free, so they do not want to leave.
2. Comparison with Alkyl Halides
- In alkyl halides (R–X), the leaving group is X⁻ (Cl⁻, Br⁻, I⁻):
- These are weak bases
- Highly stable ions
Consequence in Reactions
- Alcohols (R–OH) and ethers (R–O–R’):
- Do not undergo SN1/SN2/E1/E2 easily
- Because –OH or –OR cannot leave directly
So they are less reactive than alkyl halides.
4. Activation of Alcohols & Ethers
To make them reactive, we convert the poor leaving group into a good one.
Common activation methods:
(A) Protonation (Acidic Medium)
- In presence of acid (like H₂SO₄ or HCl):
- Now leaving group = H₂O (water)
- Water is neutral and stable → good leaving group
Conversion to Better Leaving Groups
- Convert –OH into:
- Tosylate (–OTs)
- Mesylate (–OMs)
These are excellent leaving groups.
(C) Reaction with HX
Summary (Exam Ready)
- HO⁻ and RO⁻ = strong bases → poor leaving groups
- Alkyl halides = weak base leaving groups → high reactivity
- Alcohols/ethers = less reactive
- Need activation (protonation or conversion) before reaction
