The line corresponding to the transition where the electron comes from an infinitely large orbit (n = ∞) to a fixed lower energy level.
🔹 What it means simply
- When an electron falls from very high energy levels (n = very large), the spectral lines get closer and closer.
- Finally, at n = ∞, they merge into one line → this is the series limit (limiting line).
🔹 Formula (Rydberg equation)
For limiting line
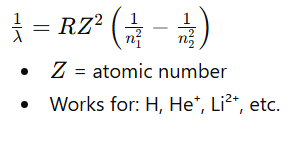
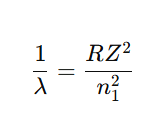
Examples of limits
- Lyman series (n₁ = 1)
- Limit: electron falls from ∞ → 1
- Region: Ultraviolet (UV)
- Balmer series (n₁ = 2)
- Limit: ∞ → 2
- Region: Visible
- Paschen series (n₁ = 3)
- Limit: ∞ → 3
- Region: Infrared
Limiting line = shortest wavelength (maximum energy) line of a series.
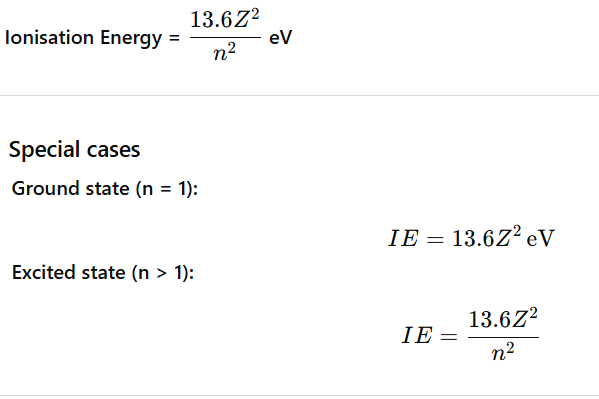
Lyman Alpha (α) Line
The Lyman α (alpha) line is the first spectral line of the Lyman series.
