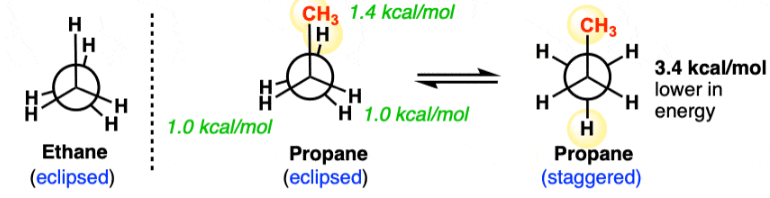
Propane (C₃H₈) shows conformational isomerism due to free rotation around the C–C single bond (σ-bond).
Key Idea
Rotation is considered around the C₁–C₂ bond (or C₂–C₃; both are equivalent).
Use Newman projection to understand conformations.
Types of Conformations in Propane
1. Staggered Conformation (Most Stable)
- Hydrogen atoms on front and back carbon are as far apart as possible
- Dihedral angle = 60°
- Minimum torsional strain
This is the lowest energy conformation
2. Eclipsed Conformation (Least Stable)
- Hydrogen atoms on front and back carbon overlap each other
- Dihedral angle = 0°
- Maximum torsional strain
This is the highest energy conformation
Important Difference from Ethane
- In propane, one carbon has CH₃ group, so:
- There are H–H eclipsing interactions
- And also H–CH₃ eclipsing interactions (more repulsion)
Hence, eclipsed propane is slightly more unstable than ethane eclipsed
Energy Profile of Propane Rotation
- Energy varies as bond rotates:
- Maxima → Eclipsed
- Minima → Staggered
There are:
- 3 staggered conformations (all equivalent)
- 3 eclipsed conformations (all equivalent)
JEE/NEET Important Points
- Only two types → staggered & eclipsed
- Staggered = most stable
- Eclipsed = least stable
- Rotation barrier ≈ 14 kJ/mol (slightly higher than ethane)
- Stability depends on torsional strain
Quick Memory Trick
“Spread = Stable, Clash = Unstable”
Why is propane eclipsed more unstable than ethane?”
Answer: Because of H–CH₃ repulsion > H–H repulsion