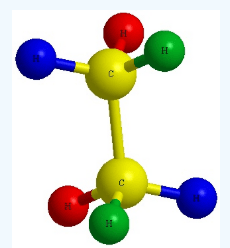
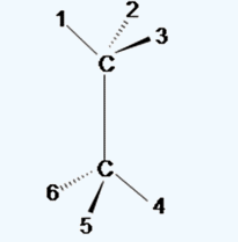
Writing wedge–dash (3D) configuration is a key skill in stereochemistry (very important for JEE/NEET). Let’s make it simple and practical.
It shows how atoms are arranged in 3D space around a carbon atom.
3 types of bonds:
- Straight line (—)
→ Bond lies in the plane of the paper - Solid wedge (▲)
→ Bond coming out toward you - Dashed wedge (▱ / —)
→ Bond going away from you
How to Draw Wedge–Dash Structure (Step-by-step)
Step 1: Identify the central atom
Usually a chiral carbon (sp³ hybridised) with 4 different groups.
Step 2: Draw two bonds in plane
Use simple straight lines for 2 groups
Step 3: Add wedge and dash
- One group → solid wedge (toward you)
- One group → dashed wedge (away from you)
Important Rules
✔ Carbon must be tetrahedral (109.5°)
✔ Always show 3D orientation clearly
✔ You can rotate the molecule, but relative positions must stay same
Interconversion Tip (Fischer → Wedge-Dash)
- Horizontal bonds in Fischer → come out (wedge)
- Vertical bonds → go back (dash)
Common Mistakes
❌ Putting all bonds in plane
❌ Using wedge/dash randomly
❌ Changing configuration while rotating
Exam Trick (JEE/NEET)
If lowest priority group (4th) is on:
- Dash (away) → read order normally
- Wedge (toward) → reverse R/S result