The E2 Reaction
MECHANISM FOR THE E2 REACTION OF AN ALKYL HALIDE
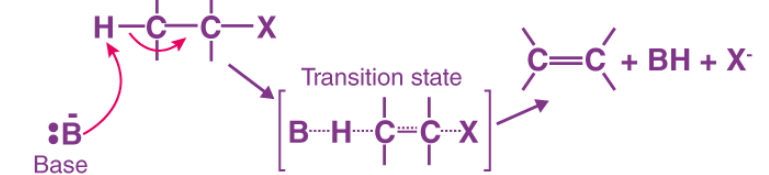
A base removes a proton (H⁺) from a carbon atom that is adjacent to the carbon bonded to the halogen. As the proton is removed, the electrons from the C–H bond move toward the neighboring carbon that is attached to the halogen. These electrons then form a double bond between the two carbons. At the same time, the halogen leaves with its bonding electron pair, because carbon cannot form more than four bonds. This process results in the formation of an alkene.
Why E2 Reaction is Regioselective
An E2 reaction is called regioselective because the elimination of hydrogen and the leaving group can produce more than one possible alkene, but one alkene is formed in greater amount than the others.
Reason
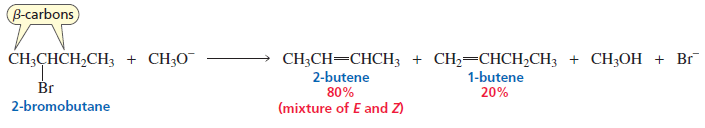
In E2 elimination, the base removes a β-hydrogen (hydrogen on the carbon adjacent to the carbon bearing the leaving group). If there are different β-carbons, the base can remove hydrogen from different positions, giving different alkenes.
However, according to Zaitsev’s rule, the more substituted alkene (the alkene with more alkyl groups attached to the double-bonded carbons) is usually more stable and therefore forms as the major product.
Zaitsev’s Rule
Alexander M. Zaitsev, a nineteenth-century Russian chemist, proposed a rule to predict the major alkene formed in elimination reactions.
According to Zaitsev’s rule, the major product is the more substituted alkene. This occurs when the base removes a hydrogen from the β-carbon that has fewer hydrogen atoms.
As a result, the double bond forms between the more substituted carbon atoms, producing the more stable alkene.
Limitations of Zaitsev’s Rule (JEE / NEET level)
Although Zaitsev’s rule usually predicts the major alkene in elimination reactions, there are several situations where it does not apply.
1. Bulky (Sterically Hindered) Bases
When a bulky base is used, it removes the most accessible hydrogen instead of the one predicted by Zaitsev’s rule.
Examples of bulky bases:
- tert-butoxide (t-BuO⁻)
- LDA
Result → Less substituted alkene (Hofmann product) becomes the major product.
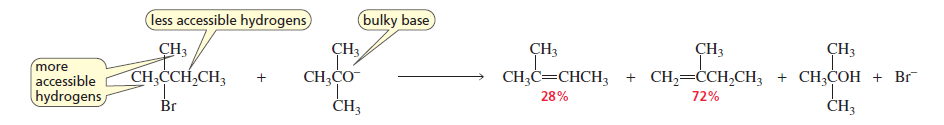
If the alkyl halide is not sterically hindered and the base is only moderately hindered, the more
stable alkene will be the major product, as expected. In other words, it takes a lot of steric hindrance
for the less stable product to be the major product. Thus, the major product of the following reaction
is 2-butene.
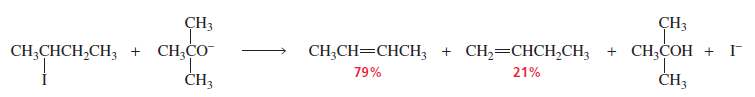

2. Poor Leaving Groups (e.g., Fluoride)
When the leaving group is F⁻, elimination often gives the less substituted alkene instead of the Zaitsev product.
Reason → Strong C–F bond and different transition state stability.
When a hydrogen and a chlorine, bromine, or iodine are eliminated from an alkyl halide, the
halogen starts to leave as soon as the base begins to remove the proton. Consequently, the transition
state resembles an alkene (see page 414).
The fluoride ion, however, is the strongest base of the halide ions and, therefore, the poorest
leaving group.
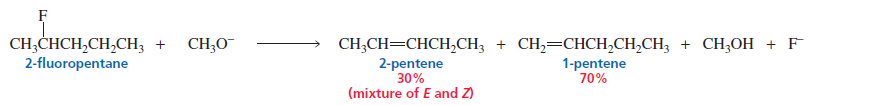
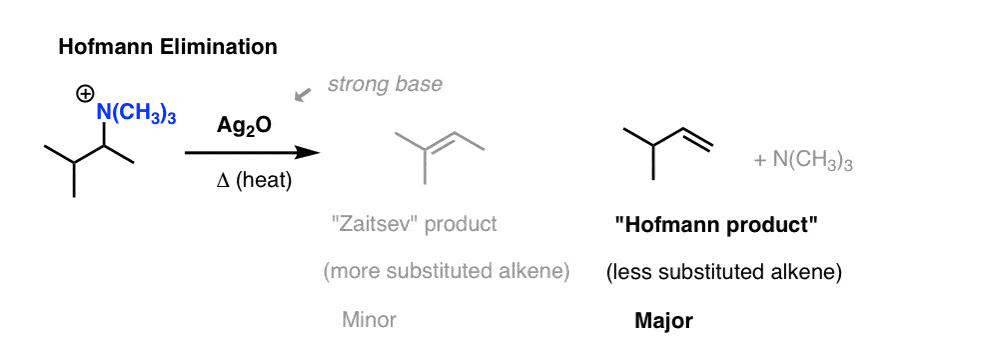
3. Quaternary Ammonium Salts (Hofmann Elimination)
Elimination of quaternary ammonium hydroxides produces mainly the less substituted alkene.
The Hofmann elimination is the preparation of alkenes from the treatment of quaternary ammonium salts with silver oxide, water, and heat.
General features: The Hofmann elimination is a β-elimination. Thus, the β-hydrogen is abstracted by the base (hydroxide ion) from the β-carbon atom. It is also an anti elimination (the leaving group has to be antiperiplanar). Besides, some side reactions occur when the base acts as a nucleophile, delivering then alcohol byproducts.
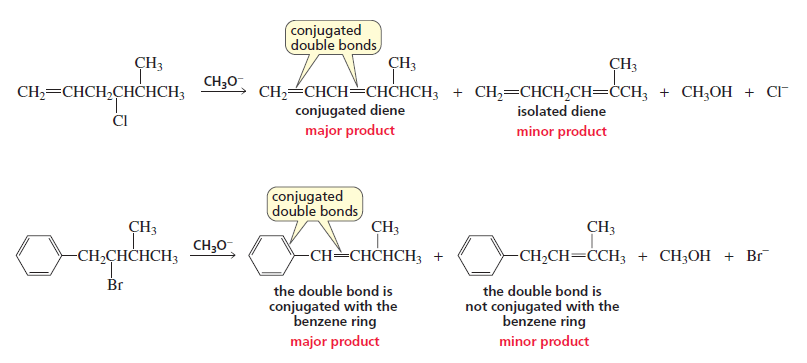
4. In each of the following reactions, the major product is the alkene with
conjugated double bonds because it is the more stable alkene, even though it is not the more
substituted alkene.
Relative Reactivities in an E2 Reaction
In E2 elimination, the rate depends on how easily the base can remove a β-hydrogen and how stable the resulting alkene will be.
Elimination from more substituted alkyl halides usually forms more substituted and more stable alkenes. Therefore, the reaction occurs more easily.
Reactivity Order of Alkyl Halides in E2
Explanation
- Tertiary alkyl halides (3°) eliminate fastest because they form highly substituted stable alkenes.
- Secondary alkyl halides (2°) react at a moderate rate.
- Primary alkyl halides (1°) react slowly because they form less substituted alkenes.
- Methyl halides (CH₃X) do not undergo E2 because there is no β-hydrogen available for elimination.
Features of E2 Reaction (JEE / NEET level)
- Bimolecular Reaction
- Two species participate: alkyl halide and base.
- Second-Order Kinetics
- One-Step (Concerted) Mechanism
- Proton removal, double bond formation, and leaving group departure occur simultaneously.
- Strong Base Required
- Examples: OH⁻, RO⁻, t-BuO⁻, NH₂⁻
- β-Elimination Reaction
- Hydrogen is removed from the β-carbon (adjacent to the carbon bearing the halogen).
- No Carbocation Intermediate
- Reaction occurs in one step, so rearrangement does not occur.
- Anti-Periplanar Requirement
- The β-hydrogen and leaving group must be in opposite (anti) orientation.
- Regioselective Reaction
- Usually follows Zaitsev’s rule → more substituted alkene forms as major product.
- Effect of Substrate Structure
Reactivity order:
3∘>2∘>1∘
- Competes with SN2 Reaction
- Strong bases with secondary or tertiary halides often favor E2 elimination.
Stereochemistry of E2 Mechanism
The E2 (Elimination Bimolecular) reaction has very important stereochemical requirements. For elimination to occur, the β-hydrogen and leaving group must have a specific spatial arrangement.
1. Anti-Periplanar Requirement
- The β-hydrogen (H) and leaving group (X) must lie in the same plane but opposite directions (180° apart).
- This arrangement is called anti-periplanar geometry.
Reason:
This alignment allows proper overlap of orbitals so the π bond of the alkene can form easily.
2. Concerted Mechanism
E2 occurs in one single step:
- Base removes β-H
- C–H bond breaks
- C–X bond breaks
- C=C double bond forms simultaneously
There is no intermediate (no carbocation).
3. Stereospecific Reaction
Because the geometry must be anti-periplanar, the reaction is stereospecific.
Example:
- Different stereoisomers of reactants can give different alkene stereoisomers (E or Z).
4. Anti Elimination
Most E2 reactions proceed by anti elimination because:
- It is energetically more stable
- Less steric repulsion between groups.
5. Syn Elimination (Rare)
- If anti arrangement is not possible, syn-periplanar elimination may occur.
- However, it is rare and higher in energy.
6. Important in Cyclic Compounds
In cyclohexane systems:
- Leaving group must be axial
- β-Hydrogen must also be axial
This creates the required anti-periplanar arrangement.
7. Product Stereochemistry
E2 generally favors formation of the more stable alkene (Zaitsev product), though bulky bases may give Hofmann product.
THE E1 REACTION
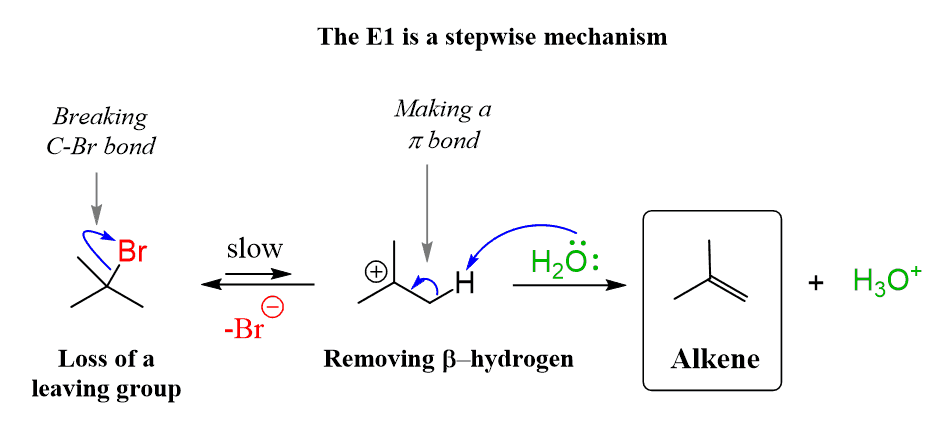
E1 Mechanism (Elimination Unimolecular) is a two-step elimination reaction commonly seen with tertiary alkyl halides in polar protic solvents. These are the main features important for JEE/NEET:
1. First-Order Kinetics
- Rate depends only on the concentration of alkyl halide.
- Rate law:
Rate=k[R–X]
2. Two-Step Mechanism
- Slow step: Leaving group leaves → carbocation formation
- Fast step: Base removes β-hydrogen → alkene formation
3. Carbocation Intermediate
- A planar carbocation is formed.
- Because of this, rearrangement (hydride shift or methyl shift) can occur.
4. Favored with Tertiary Alkyl Halides
Carbocation stability determines reactivity:
3° > 2° >> 1°
Primary carbocations are unstable, so E1 rarely occurs with primary halides.
5. Polar Protic Solvent Favours E1
Solvents like:
- H₂O
- Alcohols (ROH)
These stabilize the carbocation and leaving group.
6. Weak Bases Are Sufficient
Strong base is not required because the slow step is carbocation formation.
7. Zaitsev Alkene Major Product
The more substituted (more stable) alkene is usually the major product.
8. Competes with SN1 Reaction
Because both involve carbocation intermediate, E1 and SN1 often occur together.
9. Rearrangement Possible
Carbocation may rearrange to form a more stable carbocation, giving unexpected products.
10. Usually Occurs at Higher Temperature
Higher temperature favors elimination over substitution.
both E1 and E2 reactions can give cis and trans alkenes, but the reason and control are different.
In E1 Reaction
- Carbocation intermediate forms (planar).
- Base can remove β-H from either side.
- Therefore both cis (Z) and trans (E) alkenes form.
- Trans usually major because it is more stable.
- Not stereospecific.
In E2 Reaction
- Reaction occurs in one step.
- Requires anti-periplanar geometry between β-H and leaving group.
- The geometry of the reactant determines the alkene formed.
- So cis or trans may form depending on the starting stereochemistry.
- Stereospecific reaction.
Stereochemistry of E1 Mechanism
The E1 (Elimination Unimolecular) reaction has different stereochemistry from E2 because it proceeds through a carbocation intermediate.
1. Planar Carbocation Intermediate
- In the first step, the leaving group (X) leaves and forms a carbocation.
- The carbocation is sp² hybridized and planar.
Because it is planar, the β-hydrogen can be removed from either side.
2. Not Stereospecific
Unlike E2, E1 is not stereospecific.
Reason:
Since the intermediate carbocation is planar, the base can remove the β-hydrogen from different orientations, giving different alkene stereoisomers.
3. Formation of E and Z Alkenes
Both E (trans) and Z (cis) alkenes can form.
However:
- Trans (E) alkene is usually the major product
- because it is more stable (less steric repulsion).
4. No Anti-Periplanar Requirement
In E1, the β-hydrogen and leaving group do not need anti-periplanar geometry.
This is because:
- the leaving group already left in the first step.
5. Rearrangement Possible
Since a carbocation intermediate exists, rearrangements such as:
- Hydride shift
- Methyl shift
can occur before elimination.
6. Zaitsev Product Formation
E1 usually follows Zaitsev rule:
- More substituted alkene is the major product
