The Sabatier–Senderens reaction is a catalytic hydrogenation reaction in which unsaturated compounds (alkenes or alkynes) are reduced to saturated compounds (alkanes) using hydrogen gas (H₂) in the presence of a metal catalyst such as Ni, Pt, or Pd.
It is named after the French chemists
- Paul Sabatier
- Jean-Baptiste Senderens
Paul Sabatier received the 1912 Nobel Prize in Chemistry for his work on catalytic hydrogenation.
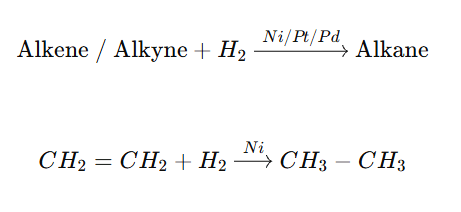
What JEE / NEET Can Ask
Concept-Based Questions
- Type of catalysis → Heterogeneous catalysis
- Catalyst used → Ni (most common), Pt, Pd
- Nature of reaction → Addition (Reduction) reaction
- Mechanism → Adsorption theory
Mechanism (Very Important for JEE)
✔ Hydrogen adsorbs on Ni surface
✔ H–H bond breaks → Atomic hydrogen formed
✔ Alkene/Alkyne adsorbs
✔ Syn addition of hydrogen
✔ Alkane formed
🔥 Very Important Concept
✔ Syn addition may give cis product
✔ But syn ≠ cis always
Example:
Hydrogenation of cycloalkene → gives cis product
But hydrogenation of 2-butene → gives alkane (no cis/trans left)
So in that case:
Syn addition happened
But cis concept disappears
Syn Addition (Mechanism term)
- Refers to how groups are added during a reaction.
- Both substituents add from the same face of the double bond.
- It is a mechanistic term.
Example:
Hydrogenation using H₂/Ni (Sabatier–Senderens) → Syn addition
What is Syn Addition?
The addition of two substituents to the same side (or face) of a double or triple bond reduces the bond order but increases the number of substituents.
Raney nickel is significantly more reactive than standard nickel powder. It is Created from a

Nickel-Aluminum alloy, where Aluminum is removed by caustic leaching (NaOH)
Platinum (like Ni, Pd) mainly prefers:
In a benzylidene compound containing a ketonic (C=O) group, when is only the C=C reduced?
Consider a Typical Example
Benzylidene acetone type compound:
Ph−CH=CH−CO−CH3
This contains:
- Aromatic ring
- C=C (alkene)
- C=O (ketone)
- Conjugation
Only Double Bond is Reduced When:
Mild catalytic hydrogenation conditions are used:
H2/Pd or Pt (room temp, low pressure)
Under these conditions:
✔ C=C reduced
✔ C=O remains intact
✔ Benzene untouched
Why C=C Reduces First?
Reactivity order:
Reasons:
- Alkene π bond adsorbs more easily on metal surface
- C=O is stabilized by resonance
- Conjugation increases stability of carbonyl
When Will C=O Also Reduce?
If you use:
- High pressure
- High temperature
- Excess hydrogen
- More active catalyst
Then both C=C and C=O may reduce.
Important JEE Rule (Remember This)
In α,β-unsaturated ketones (benzylidene type compounds):
Catalytic hydrogenation (mild) → 1,4-reduction (C=C reduction)
Strong hydride reagents (NaBH₄) → 1,2-reduction (C=O reduction)
Can NaBH₄ reduce a double bond (C=C)?
Short Answer:
What NaBH₄ Actually Reduces
NaBH₄ is a mild hydride donor.
It reduces:
✔ Aldehydes (–CHO) → 1° alcohol
✔ Ketones (>C=O) → 2° alcohol
Practical Reactivity Comparison of Common Reducing Agents (JEE Main + Advanced level)
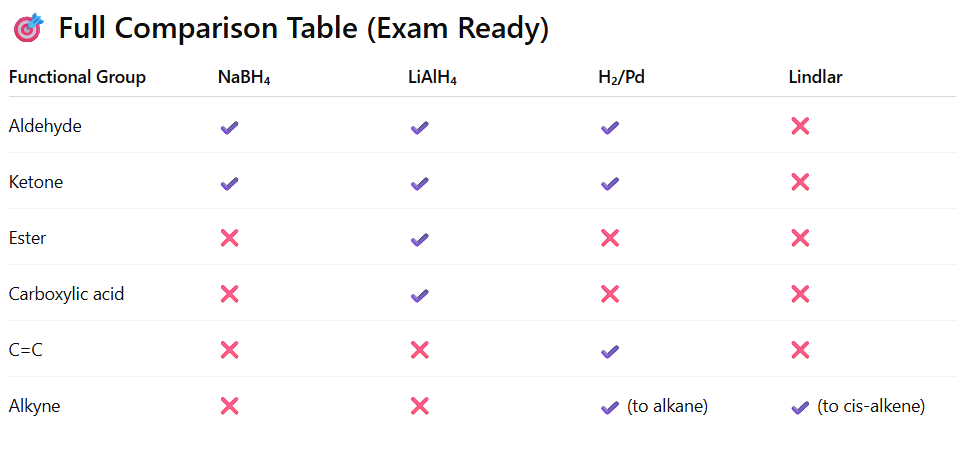
NaBH₄ (Sodium borohydride)
✔ Reduces:
- Aldehydes → 1° alcohol
- Ketones → 2° alcohol
❌ Does NOT reduce:
- C=C
- C≡C
- Carboxylic acids
- Esters
- Amides
- Benzene ring
📌 Mild, selective carbonyl reducer
📌 Works in alcohol solvent
LiAlH₄ (Lithium aluminium hydride)
✔ Reduces:
- Aldehydes
- Ketones
- Esters → 1° alcohol
- Carboxylic acids → 1° alcohol
- Amides → amines
- Acid chlorides
❌ Does NOT reduce:
- C=C
- Benzene ring
📌 Very strong hydride donor
📌 Requires dry ether
H₂ / Ni, Pd, Pt (Catalytic hydrogenation)
✔ Reduces:
- C=C
- C≡C
- Aldehydes (under suitable conditions)
- Ketones (under stronger conditions)
❌ Usually does NOT reduce:
- Esters (mild conditions)
- Benzene ring (needs high pressure)
📌 Works by surface adsorption
📌 Prefers C=C over C=O
Lindlar Catalyst
✔ Reduces:
- Alkyne → cis-alkene only
❌ Does NOT reduce:
- Further to alkane
- Carbonyl
📌 Very selective
Na / NH₃ (Dissolving metal reduction)
✔ Reduces:
- Alkyne → trans-alkene
NOTE : Reduction is highly stereoselective , giving predominantly cis -isomer
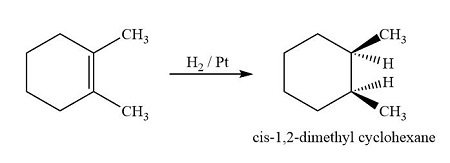
NOTE : Pd isomerises the alkene hence with Pd , trans -isomer predominates.
Palladium can catalyze alkene isomerisation via reversible adsorption on its surface, leading to thermodynamic control where the more stable trans isomer predominates.
Note: When Cyclopropane/butane reacts with hydrogen gas in the presence of Ni, it gives propane/butane
he ring opens due to high strain and behave like an alkene
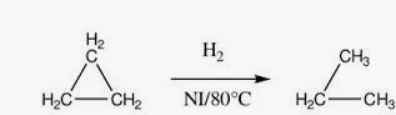
Cyclobutane also has ring strain (90° vs 109.5°)
Less than cyclopropane but still significant
Ring opens under catalytic conditions
Important JEE/NEET Concept
Reactivity order due to ring strain:
Cyclopropane> Cyclobutane > Cyclopentane
Cyclopentane and cyclohexane usually do NOT open easily.
