In decarboxylation of sodium salts of carboxylic acids, CaO (calcium oxide) is used along with soda lime.
What is Soda Lime?
Soda lime = NaOH + CaO
CaO itself does not take part directly in the reaction but plays an important supporting role.
Reaction
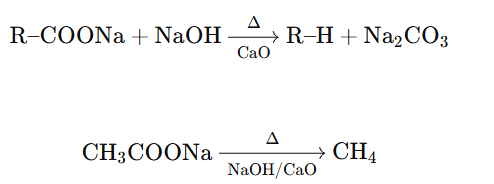
Role of CaO
- Acts as a catalyst.
- Keeps the mixture dry (dehydrating agent).
- Prevents fusion (lumping) of NaOH during heating.
- Provides porous surface for smooth reaction.
It does not get consumed in the reaction.
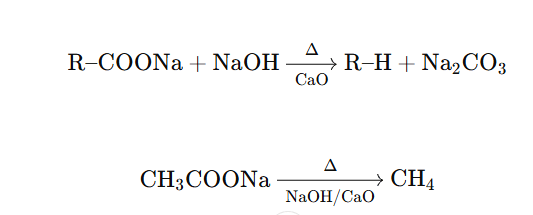
Decarboxylation proceeds via formation of a carbanion intermediate.
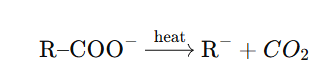
Therefore:
- More stable carbanion → easier decarboxylation
- Electron-withdrawing groups increase rate
Decarboxylation is easier when the resulting carbanion is stable:
Methyl carbanion is slightly more stable than higher alkyl due to less +I effect.
Only sodium salts undergo smooth decarboxylation.
Free acids do not give the same clean reaction under these conditions.
f soda lime is absent and only NaOH is used:
Reaction still occurs, but less efficient due to fusion of NaOH
