
Mechanism (Free Radical / SET Mechanism)

Intramolecular Wurtz Reaction (Cyclic Formation)
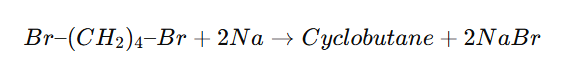
JEE / NEET Important Points
- Works best with primary alkyl halides
- Secondary & tertiary → elimination side reactions
- Dry ether is essential
- Same alkyl halide → symmetrical alkane
- Different alkyl halides → mixture of products
- Free radical intermediate
- SET (Single Electron Transfer) mechanism
- Even-numbered carbon chain formed
- Not suitable for unsymmetrical alkane synthesis
- Intramolecular reaction gives cyclic alkane
Important Concept
✔ Only alkali metals (Na, K, Li) give Wurtz-type coupling
✔ Sodium is preferred due to controlled reactivity
✔ Mg and Zn mainly form organometallic intermediates instead of direct alkane
Wurtz Reaction – Metals Other Than Sodium
Although sodium (Na) is the classical metal used in Wurtz reaction, some other metals can also promote coupling of alkyl halides
Potassium (K)
✔ More reactive than sodium
✔ Reaction is more vigorous
❌ Harder to control
📌 Rarely used in practice (safety issue)
Lithium (Li)
✔ Can participate in similar coupling
✔ Often forms organolithium intermediates
📌 Used more in organometallic synthesis than classical Wurtz
Silver (Ag)
✔ Used mainly for radical formation studies
❌ Not common for alkane synthesis
📌 Often used in rearrangement or carbocation studies
Zinc (Zn)
Zinc does not give classical Wurtz coupling easily but forms:
✔ Forms organozinc compounds
✔ Used in coupling reactions (e.g., Reformatsky type)
Magnesium (Mg)
R–X+Mg→R–MgX
✔ Forms Grignard reagent
❌ Does NOT directly give Wurtz alkane
📌 Very important alternative pathway
Cyclic Wurtz Reaction (Intramolecular Wurtz)
Cyclic Wurtz reaction is an intramolecular version of the Wurtz reaction in which a dihaloalkane reacts with sodium metal in dry ether to form a cycloalkane.
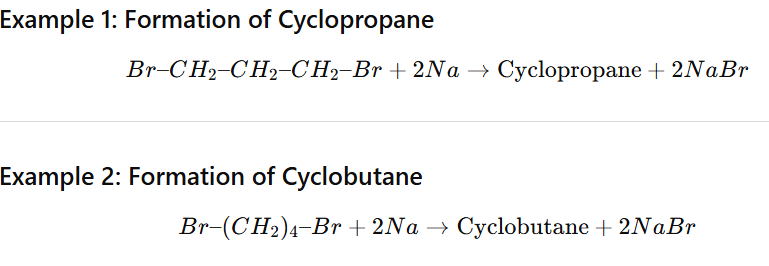
JEE / NEET Important Points
- Best for small rings (3–5 members).
- Large rings are difficult due to entropy factor.
- Competes with intermolecular Wurtz (polymerization possible).
- Primary dihalides give better yield.
- Dry ether is essential.
- Mechanism involves free radicals.
Product of Tertiary Alkyl Halide in Wurtz Reaction
When a tertiary alkyl halide (3° RX) is treated with sodium (Na) in dry ether, it does not give Wurtz coupling product efficiently.
Major product = Alkene (Elimination product)
Coupling product is minor or negligible.
Example
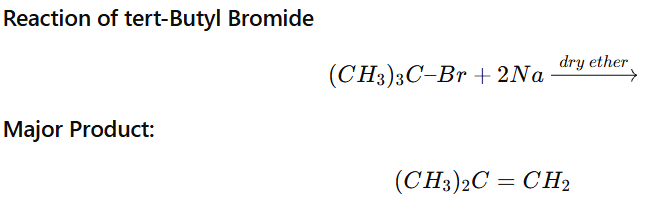
Why Elimination Occurs?
✔ Tertiary halides easily form stable tertiary radicals / carbanions
✔ Strong base character of sodium promotes β-elimination
✔ Steric hindrance prevents effective coupling
Thus, E2-type elimination dominates over coupling.
JEE / NEET Important Points
- Primary RX → best for Wurtz coupling
- Secondary RX → mixture
- Tertiary RX → alkene (major)
- Elimination increases with substitution.
- Reaction follows radical pathway but elimination competes strongly.
Reactivity Order (Bond Strength Basis)
JEE / NEET Important Points
- Alkyl bromides are preferred.
- Primary halides give best results.
- Aryl halides do NOT undergo normal Wurtz (need Wurtz–Fittig).
- Fluorides are practically inactive.
- Dry ether is essential for the reaction.
Reasons for Using Dry Ether
1️⃣ Provides Anhydrous Medium
✔ Sodium reacts violently with water:2Na+2H2O→2NaOH+H2↑
If moisture is present, sodium will react with water instead of alkyl halide.
Therefore, ether must be dry.
