Intext Questions
1.1 Calculate the mass percentage of benzene (C6H6) and carbon tetrachloride (CCl4) if 22 g of benzene is dissolved in 122 g of carbon tetrachloride.
Mass of benzene (C₆H₆) = 22 g
Mass of carbon tetrachloride (CCl₄) = 122 g
Step 1: Calculate total mass of solution
Total mass=22+122=144 g
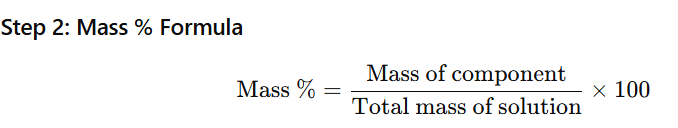
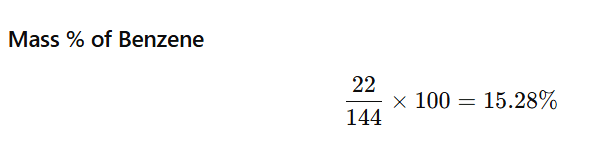

1.2 Calculate the mole fraction of benzene in solution containing 30%
by mass in carbon tetrachloride
Solution contains 30% by mass benzene in carbon tetrachloride (CCl₄)
That means in 100 g of solution:
- Mass of benzene (C₆H₆) = 30 g
- Mass of carbon tetrachloride (CCl₄) = 70 g
Step 1: Calculate molar masses
- Molar mass of C₆H₆ = g/mol
- Molar mass of CCl₄ = g/mol
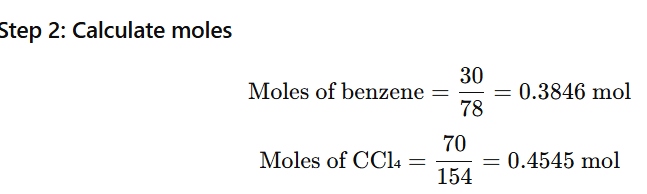

1.3 Calculate the molarity of each of the following solutions: (a) 30 g of
Co(NO3)2. 6H2O in 4.3 L of solution (b) 30 mL of 0.5 M H2SO4 diluted to
500 mL
Calculate molar mass
For Co(NO₃)₂·6H₂O
- Co = 59
- (NO₃)₂ = 2 × (14 + 3×16) = 2 × 62 = 124
- 6H₂O = 6 × 18 = 108
Molar mass=59+124+108=291 g/mol
Step 2: Calculate moles
Moles=30/291=0.103 mol

b) 30 mL of 0.5 M H₂SO₄ diluted to 500 mL
Use dilution formula:
Given:
- M
- mL
- mL
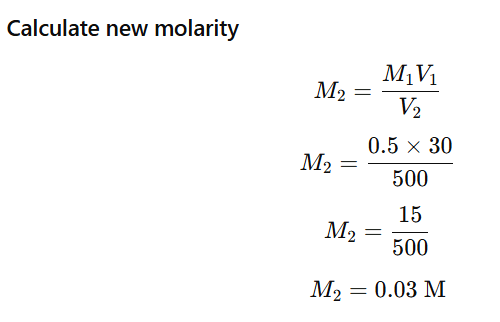
1.4 Calculate the mass of urea (NH2CONH2) required in making 2.5 kg of
0.25 molal aqueous solution
Mass of solution = 2.5 kg
Molality (m) = 0.25 mol/kg
Solute = Urea (NH₂CONH₂)
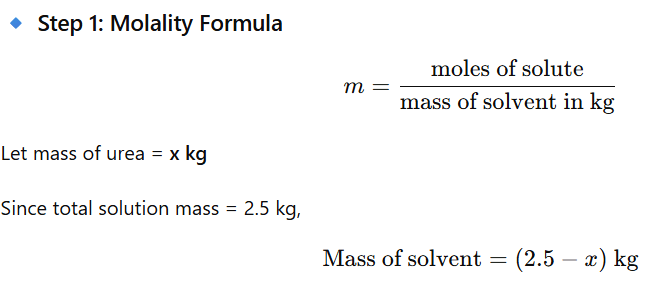
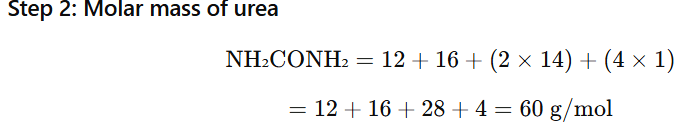

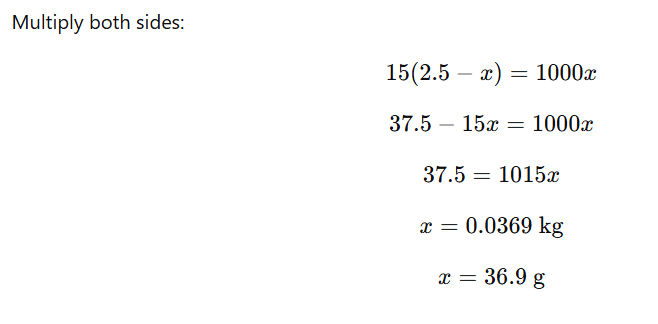
1.5 Calculate (a) molality (b) molarity and (c) mole fraction of KI if the density
of 20% (mass/mass) aqueous KI is 1.202 g mL-1
20% (w/w) aqueous KI
Density = 1.202 g mL⁻¹
That means:
In 100 g of solution
- Mass of KI = 20 g
- Mass of water = 80 g
Molar mass of KI =



1.6 H2S, a toxic gas with rotten egg like smell, is used for the qualitative analysis. If the solubility of H2S in water at STP is 0.195 m, calculate Henry’s law constant.
To calculate Henry’s law constant (Kₕ), we use:
p=KH×x
Where:
- = partial pressure of gas (at STP = 1 atm)
- = mole fraction of gas in solution
- Given solubility = 0.195 molal (m)
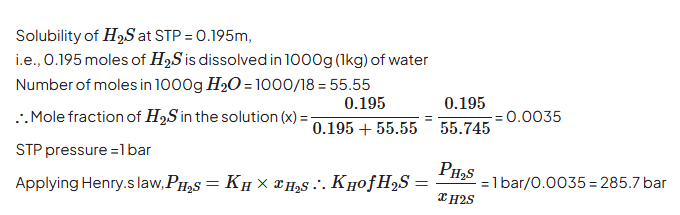
1.7 Henry’s law constant for CO2 in water is 1.67×108 Pa at 298 K. Calculate
the quantity of CO2 in 500 mL of soda water when packed under 2.5 atm
CO2 pressure at 298 K.
Given:
- Henry’s constant,
- Pressure of CO₂ = 2.5 atm
- Volume of soda water = 500 mL = 0.5 L
- Temperature = 298 K
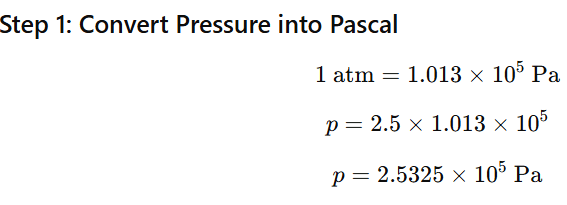
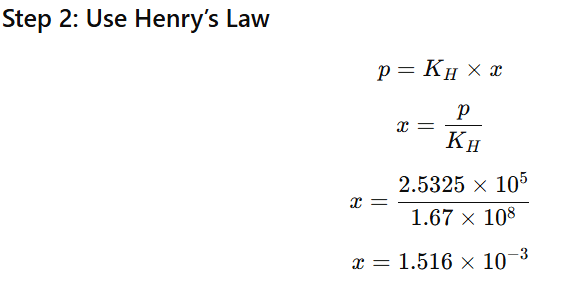
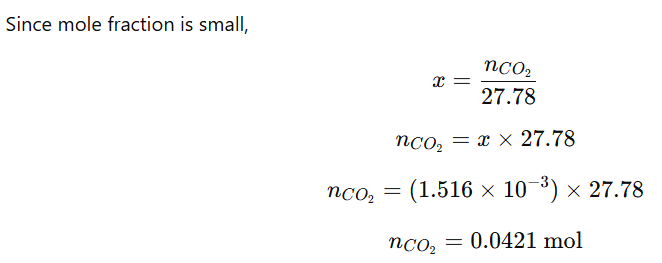
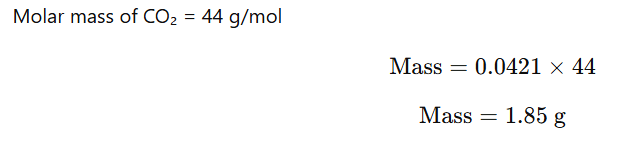
1.8 The vapour pressure of pure liquids A and B are 450 and 700 mm Hg
respectively, at 350 K . Find out the composition of the liquid mixture if total
vapour pressure is 600 mm Hg. Also find the composition of the vapour phase.
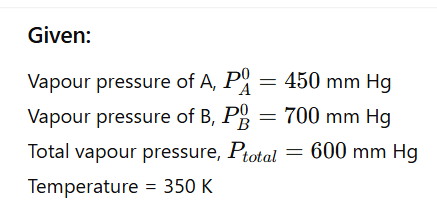


1.9 Vapour pressure of pure water at 298 K is 23.8 mm Hg. 50 g of urea (NH2CONH2) is dissolved in 850 g of water. Calculate the vapour pressure of water for this solution and its relative lowering.
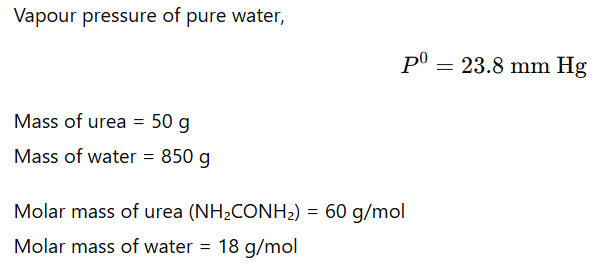
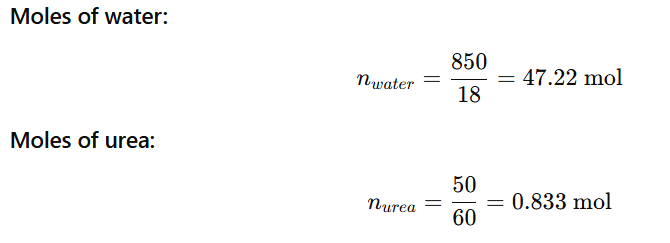
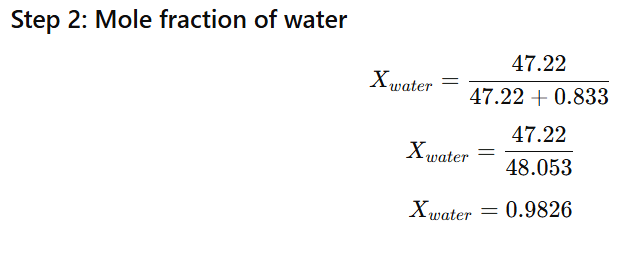
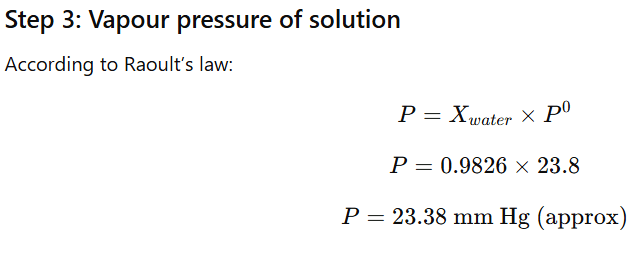

1.10 Boiling point of water at 750 mm Hg is 99.63°C. How much sucrose is to be added to 500 g of water such that it boils at 100°C.
Given:
Boiling point at 750 mm Hg = 99.63°C
Required boiling point = 100°C
ΔTb=100−99.63=0.37∘C
Mass of water = 500 g = 0.5 kg
For water: Kb=0.52Kkgmol−1
Sucrose is non-electrolyte →
Molar mass of sucrose (C₁₂H₂₂O₁₁) = 342 g/mol


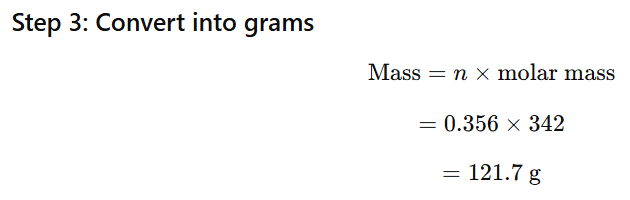
1.11 Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolved in 75 g of acetic acid to lower its melting point by 1.5°C. Kf = 3.9 K kg mol-1
Given:
Depression in freezing point: ΔTf=1.5∘C
Kf=3.9Kkgmol−1
Mass of solvent (acetic acid) = 75 g = 0.075 kg
Ascorbic acid (C₆H₈O₆) is non-electrolyte →
Molar mass of ascorbic acid:
6(12)+8(1)+6(16)
=72+8+96=176g/mol
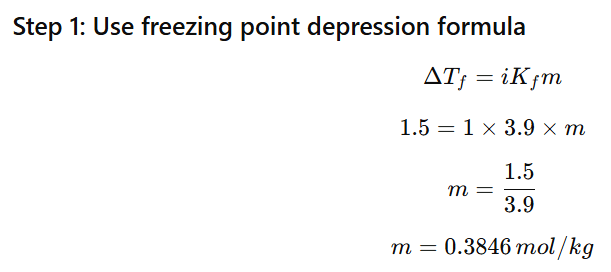
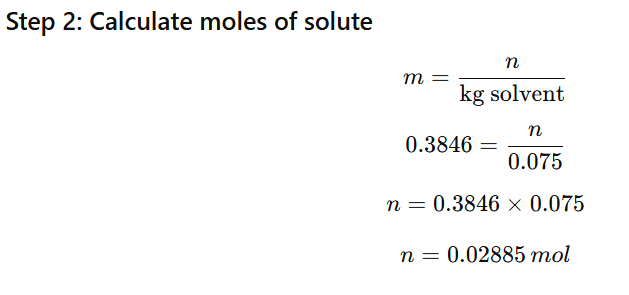
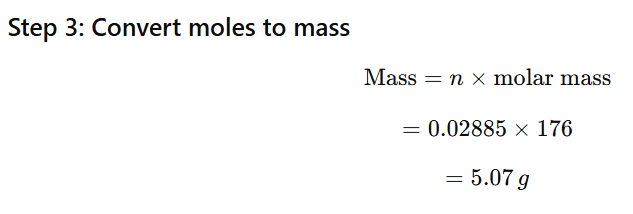
1.12 Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at 37°C
Given:
Mass of polymer = 1.0 g
Molar mass = 185,000 g/mol
Volume of solution = 450 mL = 0.450 L
Temperature = 37°C = 310 K
Gas constant:
R=0.0821Latmmol−1K−1
Step 1: Use osmotic pressure formula
π=CRT
First find molarity (C).

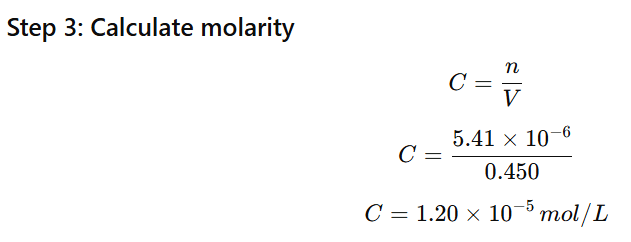
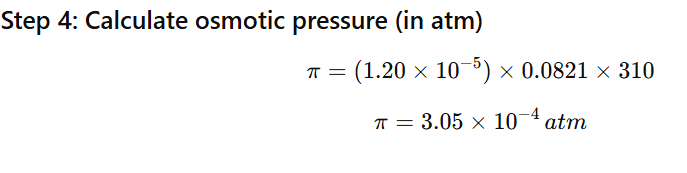
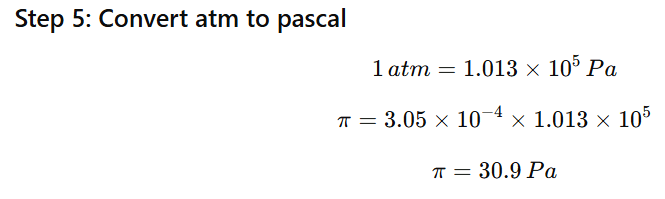
Exercises
1.1 Define the term solution. How many types of solutions are formed? Write briefly
about each type with an example.
Definition of Solution
A solution is a homogeneous mixture of two or more components in which:
- The component present in larger amount is called solvent
- The component present in smaller amount is called solute
Example: Salt dissolved in water.
Types of Solutions
Solutions are classified on the basis of physical state of solute and solvent.
There are 9 types of solutions:
1. Gas in Gas Solution
- Solvent: Gas
- Solute: Gas
- Example: Air (O₂, CO₂ dissolved in N₂)
2. Gas in Liquid Solution
- Solvent: Liquid
- Solute: Gas
- Example: CO₂ in water (Soda water)
3. Gas in Solid Solution
- Solvent: Solid
- Solute: Gas
- Example: Hydrogen in palladium
4. Liquid in Gas Solution
- Solvent: Gas
- Solute: Liquid
- Example: Moist air (water vapour in air)
5. Liquid in Liquid Solution
- Solvent: Liquid
- Solute: Liquid
- Example: Alcohol in water
6. Liquid in Solid Solution
- Solvent: Solid
- Solute: Liquid
- Example: Dental amalgam (Mercury in silver)
7. Solid in Gas Solution
- Solvent: Gas
- Solute: Solid
- Example: Smoke (dust particles in air)
8. Solid in Liquid Solution
- Solvent: Liquid
- Solute: Solid
- Example: Salt in water
9. Solid in Solid Solution
- Solvent: Solid
- Solute: Solid
- Example: Alloys (Brass = Zn in Cu)
1.2 Give an example of a solid solution in which the solute is a gas.
Hydrogen gas dissolved in palladium metal is an example of a solid solution in which the solute is a gas.
1.3 Define the following terms:
(i) Mole fraction (ii) Molality (iii) Molarity (iv) Mass percentage
