Surface chemistry
Q.1.Define Adsorption.
Ans.The accumulation of molecules of a substance on the surface of solid is called adsorption.
Q.2.Define adsorbent and adsorbate.
Ans. Substance, adsorbed on the surface is called the adsorbate and the substance on which adsorption takes place is called adsorbent.
Q.3.Define desorption.
Ans.The removal of the adsorbed substance from the surface of adsorbent is called desorption.
Q.4.Define absorption.
Ans.The process in which the substance is uniformly distributed throughout the body of material is called absorption.
Q.5.Define sorption.
Ans. When both adsorption and absorption take place simultaneously.
Q.6.Give some examples of adsorption.
Ans.1. Adsorption of gases like O2, H2, CO, Cl2, NH3 or SO2 on the surface of charcoal.
2.Colouring substances are adsorbed by the animal charcoal.
3. Moisture get adsorbed on the surface of the alumina or silica gel.
Q.7. It is observed that the pressure of the closed vessel Containing O2, H2, CO, Cl2, NH3 or SO2 decreases when charcoal is kept in it.
Ans .It is due to Adsorption of gases like O2, H2, CO, Cl2, NH3 or SO2 on the surface of charcoal.
Q.8.When aqueous solution of raw sugar passed over beds of animal charcoal, becomes colourless why?
Ans. It is due to coloring substances are adsorbed by the animal charcoal.
Q.9. The air becomes dry in the presence of silica gel why?
Ans. It is due to moisture get adsorbed on the surface of the alumina or silica gel.
Q.10. When animal charcoal is added in a solution of an organic dye it becomes colourless why?
Ans. The molecules of the dye adsorbed on the surface of charcoal.
Q11.Explain the mechanism of adsorption.
Ans. on the surface the particles are not surrounded by atoms or molecules of their kind on all sides, and hence they possess unbalanced attractive forces towards downward direction.
Adsorption can also explained by thermodynamics principle.
1.ΔH(Enthalpy change) in adsorption is negative. During adsorption there is always a decrease in residual forces of the surface. That is there is deceases in surface energy which appears as heat.
2. ΔS(Entropy change) in adsorption is negative because in adsorption the freedom of movement of adsorbate molecules become restricted.
Hence according to ΔG = ΔH – TΔS, ΔG can be negative if ΔH has sufficiently high negative value as – TΔS is positive
Q.12. Explain types of Adsorption.
Ans. There are two types of adsorption of gases on solids
1.Physisorption
2.Chemisorption
1.Physisorption
If adsorption of gas on the surface of a solid occurs on account of weak van der Waals’ forces, the adsorption is called physisorption.
2. Chemisorption
When the gas molecules or atoms are held to the solid adsorbent surface by chemical bonds (covalent or ionic), the adsorption is called chemisorption.
Q.13. What do you mean by specificity in physisorption?
Ans In physisorption a given surface of an adsorbent does not show any preference for a particular gas as the van der Waals’ forces are universal.
Q.14.How does physisorption depends on nature of adsorbate:
Ans.Easily liquefiable gases with higher critical temperatures are readily adsorbed .
Q.15.Arrange the amount of adsorption of methane , sulphur dioxide and hydrogen on 1g of activated charcoal oorder.
Ans. Sulphur dioxide (SO2)>Methane(CH4)> Hydrogen(H2).
It is due to critical temperature decreases in following order
Sulphur dioxide (SO2)>Methane(CH4)> Hydrogen(H2)
Q.16. Explain the reversible nature of physisorption.
Ans. Adsorbtion increases when pressure is increased as the volume of the gas decreases according to Le–Chateliers’s principle and the gas can be removed by decreasing pressure thus physisorption is reversible in nature .
Q.17.The physisorption occurs readily at low temperature and decreases with increasing temperature why.
Ans. Since adsorption is exothermic reversible process hence it goes towards backward direction on increasing temperature according to Le-Chatelier’s principle.
Q.18.How does adsorption depends on the surface area of adsorbent explain.
Ans.The amount of adsorption increases with the increase in surface area of the adsorbent.
Q19. Finely divided metals and porous substances are good adsorbents why.
Ans.It is due to their large surface area finely divided metals and porous substances are good adsorbents.
Q.20. Why enthalpy of physisorption is quite low.
Ans. It is due to the attraction between adsorbent and adsosrbate molecules is only weak van der Waals’ forces.
Q.21. What do you mean by high specificity of chemisorption.
Ans. Chemisorption is highly specific and it will only occur if there is some possibility of chemical bonding between adsorbent and adsorbate.
Q.22. Explain the Irreversible nature of chemisorption.
Ans.Since chemisorptions involves chemical bond formation hence it is Irreversible in nature .
Q.23 Chemisorption occurs at higher temperature and increases with increasing temperature why.?
Ans. Since chemisorption involves chemical bond formation hence adsorbate molecules need energy of activation. More number of molecules get required amount of activation energy when temperature increases hence chemisorption increases on increasing temperature.
Q.24. Enthalpy of chemisorption is high why?
Ans.Since chemisorption involves chemical bond formation hence enthalpy of chemisorption is high.
Q.25.Why physisorption is multimolecular and chemisorption is unimolecular.
Ans. Since chemisorption involves chemical bond formation and in physisorption adsorbate molecules are attracted by adsorbent by van der Waals’ forces.
Q.26.Define adsorption isotherm.
Ans. The relation in the amount of gas adsorbed by the adsorbent with pressure at constant temperature can be expressed by means of a curve known as adsorption isotherm.
Q.27.Explain Freundlich adsorption isotherm.
Ans. The relation between the quantity of gas adsorbed by unit mass of solid adsorbent and pressure at a particular temperature can be expressed by the following equation called Freundlich adsorption isotherm
x/m= k.p1/n (n > 1)
Where x= mass of the gas adsorbed.
m= mass m of the adsorbent.
P =Pressure
k and n = constants.
Taking logarithm on both sides.
log(x/m)= log k + 1/n log p.
Q.28.When adsorption is independent of pressure and when it is directly proportional to the pressure.
Ans.Minimum Value of 1/n is o and maximum value is 1
On minimum value
log(x/m)= log k + 0.
log p
log(x/m)= log K
log(x/m)= constant
hence adsorption is independent of pressure.
On maximum value
log(x/m)= log K + 1.
log p
log(x/m)= log K+ logp
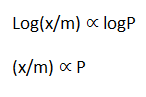
Adsorption is directly proportional to the pressure.
Q.29.What are the applications of adsorption.
Ans. (i) Production of high vacuum.
(ii) Gas masks.
Charcoal is used as adsorbent in gas masks.
(iii)Controlling humidity.
Silica gel and alumina gel can adsorbs the moisture.
(iv) Removal of colouring matter from solutions.
Animal charcoal can adsorbs colouring matter from solutions
(v) Heterogeneous catalysis.
During Heterogeneous catalysis catalyst adsorbs the reactant molecules on surface of it.
(vi) Separation of inert gases
Due to the difference in degree of adsorption by different inert gases.
(vii) In curing diseases
(viii) Froth floatation process
(ix) Adsorption indicators
(x)In chromatographic analysis.
Q.30.Define catalyst.
Ans.Substances, which increase the rate of a chemical reaction and remain chemically and quantitatively unchanged after the reaction, are called catalysts.
Q.31.Define Promoters and poisons.
Ans. Substances that enhance the activity of a catalyst are called promoters.
In Haber’s process for manufacturing ammonia, molybdenum acts as a promoter for iron catalyst.
And substances that decrease the activity of a catalyst are called poisons.
Q.32.Explain the type of catalysis.
Ans. Catalysis can be divided into two groups.
- Homogeneous catalysis.
- Heterogeneous catalysis.
Homogeneous catalysis.
When the reactants and the catalyst are in the same phase the process is called homogeneous catalysis.
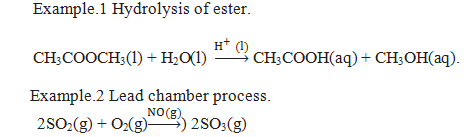
Heterogeneous catalysis.
When the reactants and the catalyst are in the different phase the process is called Heterogeneous catalysis.
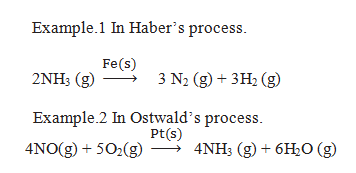
Q.33. Explain the mechanism of heterogeneous catalysis.
Ans. (i) Diffusion of reactants to the surface of the catalyst.
(ii) Adsorption of reactant molecules on the surface of the catalyst.
(iii) Chemical reaction between reactants on the surface of catalyst and formation of an intermediate
(iv) Desorption of reaction products from the catalyst surface, and therefore making the surface available again for more reaction to occur.
(V) Diffusion of reaction products away from the catalyst’s surface.
Q.34.What do you mean by activity of catalyst.
Ans.It must be strong enough to make the catalyst active whereas, not so strong that the reactant molecules get immobilized on the catalytic surface leaving no further space for the new reactants to get adsorbed.
Q.35.What do you mean by selectivity of catalyst.
Ans. A particular substance can catalyse only a particular reaction. A substance which catalyse one reaction may fail to catalyse another reaction.
Q.36.Define shape-selective catalysts.
Ans.The catalytic reaction that depends upon the pore structure of the catalyst and the size of the reactant and product molecules is called shape-selective catalysis.
Example. Zeolites.
ZSM-5 a Zeolites It converts alcohols into gasoline (petrol)
Q.37.Define Enzyme Catalysis.
Ans. Reactions that occur in the bodies of animals and plants to maintain the life process are catalysed by enzymes such reactions are called enzyme Catalysis.
Examples.
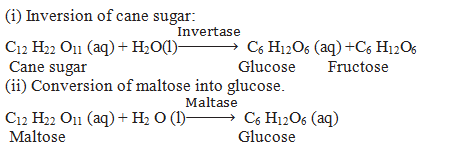
(iii) Conversion of milk into curd
Lacto bacilli enzyme present in curd converts milk into curd
(iv) The pepsin enzyme present in stomach, converts proteins into peptides and in intestine, the pancreatic trypsin converts proteins into amino acids .
Q.38.What are the characteristics of enzyme catalyst.
Ans. (i)highly efficient
(ii) Highly specific: Each enzyme is specific for a particular reaction.One catalyst cannot catalyse more than one enzymic reaction
(iii) At optimum temperature enzymes are highly active.The rate of an enzyme reaction is maximum at a particular temperature, called the optimum temperature. On either side of the this optimum temperature, the decreases. Human body optimum temperature is 310K
(iv) At optimum pH enzymes are highly active.
The rate of an enzyme reaction is maximum at a particular pH, called the optimum pH. On either side of the this optimum pH, the activity of enzyme decreases
(v) Activators and co-enzymes Increase the activity of enzyme.
activators and co-enzymes both increase the activity of catalyst but activators are metal ions such as Na+, Mn2+, Co2+, Cu2+and co-enzymes are other substances.
(vi) Effects of inhibitors and poisons
Substances which mimic or destroy the catalytic activity of the enzymes completely called inhibitors and poisons.
Example drugs.
Q.39.When a small non-protein (vitamin) is present along with an enzyme, the catalytic activity is enhanced considerably why.
Ans.It is due to non-protein (vitamin) works like co-enzyme for enzyme catalyst and increase the activity of catalyst.
Q.40. Amylase in presence of sodium chloride i.e., Na+ ions are catalytically very active why.?
Ans. It is due to Na+ ions work like activators for enzyme catalyst and increase the activity of catalyst.
Q.41.Explain the Mechanism of enzyme catalysed reaction.
Ans. Step 1: Enzyme catalyst binds with substrate to form an activated complex.
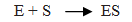
Step 2: Activated complex decomposed and form product.
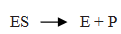
Q.42.Define colloids.
Ans.A heterogenous mixture of two components in diameter of dispersed phase particle is 1 to1000 nm .
Q.43.Classify colloids on the basis of physical state of dispersed phase and dispersion medium.
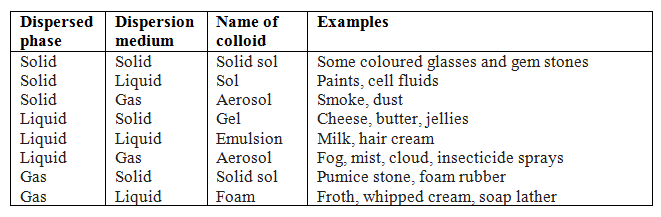
Q.44.Define aquasol or hydrosol.
Ans.Types of sol(solid dispersed into liquid dispersion medium) in which water acts as dispersion medium.
Q.45.Define alcosol.
Ans.Types of sol(solid dispersed into liquid dispersion medium) in which alcohol acts as dispersion medium.
Q.46. Classify colloids on the basis of nature of interaction between the dispersed phase and the dispersion medium.
Ans.There are two types of colloids on the basis of nature of interaction between the dispersed phase and the dispersion medium.
(i) Lyophilic colloids.
(ii) Lyophobic colloids.
(i) Lyophilic colloids.
The type of colloid which is formed by simple mixing of dispersed phase into suitable dispersion medium called Lyophilic colloids .Example: When gum, gelatine, starch, rubber, etc. is dissolved into suitable medium.
(ii) Lyophobic colloids.
The type of colloid which can’t be formed by simple mixing of dispersed phase into suitable dispersion medium but only by special methods called Lyophobic colloids.
Examples.When metal or their sulphides is dissolved into suitable medium.
Q.45. Why Lyophilic colloids are called reversible sols.
Ans. If the dispersion medium is separated from the dispersed phase the sol can be reconstituted by simply remixing with the dispersion medium.
Q.46. Why Lyophilic colloids are quite stable and cannot be easily coagulated.?
Ans.In lyophilic colloid ,particles have great interaction with dispersion medium hence Lyophilic colloids are quite stable and cannot be easily coagulated.
Q.47. Why Lyophobic colloids are called irreversible sols.
Ans.Once the dispersion medium is separated from the dispersed phase the sol cannot be reconstituted by simple addition of the dispersion medium.
Q.48.Why lyophobic sols need stabilizing agents for their preservation.
Ans.Lyophobic colloids are not quite stable and can be easily coagulated hence it needs stabilizing agents for their preservation.
Q.49.Classify colloids on the basis of type of the particles of the dispersed phase.
Ans. There are three types of colloids on the basis of type of the particles of the dispersed phase.
- Multimolecular colloids
- Macromolecular colloids
- Associated colloids
(i) Multimolecular colloids:
When smaller molecules (which size is not in colloidal range) present in suitable dispersion medium aggregate together to form species having size in the colloidal range form a colloids called multimolecular colloids.
Macromolecular colloids:
These types of colloid are formed by natural macromolecules ike starch, cellulose, proteins and enzymes or by man made macromolecules like polythene, nylon, polystyrene, synthetic rubber when dissolved in suitable dispersion medium.
(iii) Associated colloids.
When soaps or sodium potassium salts of long of a higher fatty acid present in suitable dispersion medium they aggregate together to form a aggregate called micelles and thus colloids are formed called associated colloids .
Q.50.Define CMC (Citical micelle concentration).
Ans. The formation of micelles takes place only above a particular concentration called CMC or critical micelle concentration .
Q.51. Define Kraft temperature (Tk) .
Ans.The formation of micelles takes place only above a particular temperature is called Kraft temperature (Tk) .
Q.52.Explain the mechanism of micelle formation.
Ans.Sodium stearate (an example of salt) is dissolved in water.It is dissociated into C17H35COO– anion and Na+ and cation. The C17H35COO– ions, contain two parts, a long hydrocarbon chain R (C17H35) called non-polar hydrophobic ‘tail’ and a polar group COO– a polar-ionic head. At lower concentration the C17H35COO– ions are present on the surface with their COO– groups in water and the hydrocarbon chains R staying away from it and remain at the surface. At CMC the anions are aggregated to form a spherical shape with their hydrocarbon chains pointing towards the centre of the sphere with COO– part remaining outward on the surface of the sphere. This aggregate is called micelle.
A.51.Explain the cleansing action of soap.
Ans.A micelle forms around grease. The hydrophobic part is in the grease and hydrophilic part projects out of the grease. Since the hydrophilic part can interact with water the oil droplet pulled in water and removed from the dirty surface.
Q.53.How can we prepare the lyophobic colloid by chemical methods give some examples.
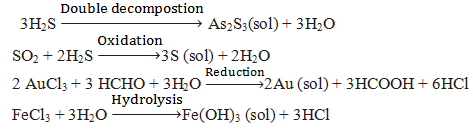
Q.54. How can we prepare the lyophobic colloid by electrical disintegration(Bredig’s Arc) method.
Ans. An ice bath is taken. A container containing sutaible dispersion medium is kept in the centre of the ice bath. An electric arc is struck between electrodes of the metal immersed in the dispersion medium.The intense heat produced vapourises the metal, which then condenses to form particles of colloidal size.
Q.55. How can we prepare the lyophobic colloid by electrical peptization method.
Ans.The process in which a freshly formed precipitate substance is shaked with suitable dispersion medium and forms colloid. A small amount of electrolyte used for this purpose is called peptizing agent.
Q.56.Explain dialysis.
Ans .Dialysis method is used for purification of colloid. In this method smaller dissolved impurities are removed out from colloidal solution by diffusion method using suitable membrane.
Q.57.Explain eletro-dialysis.
Ans.Simple dialysis is very slow, It can be made faster by applying an electric field in the impure colloidal solution.The ions present in the colloidal solution migrate out to the oppositely charged electrodes.
Q.58.Expain the ultrafiltration method for the preparation of colloidal solution.
Ans. Specially prepared filter called ultrafilter is used, This filter permit all substances to pass except the colloidal particles through its pores, this method of purification is called ultrafiltration.
Q.59.Explain some properties of colloid.
Ans.(i) Colligative properties.
They show all types of colligative properties that is relative lowering of vapour pressure,
depression in freezing point, osmotic pressure and elevation in boiling point.
Q.60.Define Tyndall effect .
Ans.The path of light becomes visible when beam of light is passed through colloidal solution. It is due to scattering of light in all directions by colloidal particles.
Q.61.Give an example of Tyndall effect.
Ans. Path of projection of picture in the cinema hall becomes visible due to scattering of light by dust and smoke particles.
Q.62.Which property of colloidal solution is used to distinguish between a colloidal and true solution.
Ans. Tyndall effect.
Q.63.Which property of colloidal solution is responsible for the colour of colloidal solution.
Ans.Tyndall effect is responsible for color of the colloids that is light scattered by the dispersed particles.
Q.64.Explain factors effecting colour of the colloids.
Ans.colour of the colloids depends on the following two factors.
1.Size of the colloidal particles.
Finest gold sol is red in colour; as the size of particles increases , it appears purple, then blue and finally golden.
2.The manner in which the observer receives the light.
A mixture of milk and water appears blue when viewed by the reflected light and red when viewed by the transmitted light.
Q.65.Define Brownian movement.
Ans.The continuous zig-zag motion of colloidal particles is called Brownian movement.
Q.64.Explain the reason of Brownian movement.
Ans.It is due to the unbalanced bombardment of the colloidal particles by the molecules of the dispersion medium.
Q.66. Which property of colloidal solution is called stirring effect which does not permit the particles to settle and thus, is responsible for the stability of sols.
Ans. Brownian movement.
Q.67.Explain the factors effecting Brownian movement.
Ans. Brownian movement depends on following factors.
1.Size of colloidal particles.
Smaller the size of colloidal particles faster is the motion.
2.viscosity.
Lesser the viscosity of colloidal particles faster is the motion.
Q.68.Give some examples of positive colloids.
Fe2O3.xH2O, etc.Basic dye stuffs, methylene blue sol.Haemoglobin (blood) Oxides, TiO2 sol.
Q.69.Give some examples of negative colloids.
Metals, e.g., copper, silver, gold sols.Metallic sulphides, e.g., As2S3,Sb2S3, CdS sols.Acid dye stuffs, e.g., eosin, congo red sols.Sols of starch, gum, gelatin,clay, charcoal, Blood etc.
Q.70.Explain he reason behind charge on colloidal particles.
Ans.Colloidal particles adsorbed the common –ve or +ve ion from the adsorption medium this method is called preferential adsorption of ions.
Q.71. When silver nitrate solution is added to potassium iodide solution, negatively charged colloidal solution(AgI/I–) is formed why.?
Ans.Because the precipitated silver iodide adsorbs common iodide ions from the dispersion medium.
Q.72.When potassium iodide solution is added to silver nitrate solution, positively charged colloidal solution(AgI/Ag+)is formed why?
Ans. Because the precipitated silver iodide adsorbs common Ag+ ions from the dispersion medium.
Q.73.When FeCl3 is added to excess of hot water, a positively charged sol of hydrated ferric oxide (Fe2O3.xH2O/Fe3+) is formed why.
Ans.It is due to adsorption of Fe3+ ions by colloidal particles Fe2O3.
Q.74.When FeCl3 is added to excess of NaOH, a negatively charged sol of hydrated ferric oxide (Fe2O3.xH2O/OH–) is formed why.
Ans. It is due to adsorption of OH– ion by colloidal particles.
Q.75. Define electrokinetic potential or zeta potential .
Ans.The potential difference between the fixed layer and the diffused layer is called electrokinetic potential or zeta potential.
Q.76.Define electrophorsis.
Ans. The experiment which confirms existence of charge on colloidal particles is called electrophoresis. In this method electric potential is applied across two platinum electrodes dipping in a colloidal solution, if the the colloidal particles contain +ve charge they move toward the -ve electrode or vice versa.
Q.77.Define electrosmosis.
Ans.The movement of the dispersion medium towards an electric field when movement of colloidal particles towards electrode in electrophorsis is prevented is called eletroosmmosis.
Q.78.Define Coagulation or precipitation.
Ans.The process of settling down of the colloidal particles is called coagulation.
Q.79.Explain mutual coagulation .
Ans.Mutual coagulation is one of the coagulation method for coagulation of the colloids.In this method two oppositelly charged sols are mixed in equal proportions, they neutralise their charges and get precipitated.
Q.80.Mixing of hydrated ferric oxide and arsenious sulphide bring them in the precipitated forms why?
Ans.Mutual coagulation occurs between hydrated ferric oxide(+ve) colloids and arsenious sulphide(-ve) and they get precipitated.
Q.81.How can colloids get coagulated on boiling.
Ans. Charge on colloidal particles is responsible for the stability of sols. On boiling increased collisions between colloidal particles and molecules of dispersion medium reduce the charge and colloids get coagulated.
Q.82.Explain Hardy-Schulze rule.
Ans.The higher the charge on –ve or +ve flocculating ion or coagulating ion which is added to the +ve or –ve colloids respectively for coagulation higher is its power of precipitation.
Examples.
Al3+>Ba2+>Na+(For –ve colloids)
[Fe(CN)6]4– > PO43– > SO42– > Cl–(For +ve colloids)
Q.83.How lyophilic colloids become protective colloids for lyophobic colloids.
Ans.Due to strong affinity of dispersed particles with dispersion medium lyophilic sols are highly stable.Thus when a lyophilic sol is added to the lyophobic sol, the lyophilic particles form a layer around lyophobic particles and thus protect the lyophobic collids from coagulating ion.
Q.84.Define emulsion.
Ans.The types of colloid in which liquid dispersed phase is dispersed into liquid dispersion medium.
Q.85.Explain the different types of emulsion.
Ans.There are two types of emulsion.
O/W emulsions(Oil in water)
Types of emulsion in which fat is dispersed phase and water is dispersion medium.
Example:Milk
W/O emulsions(water in oil)
Types of emulsion in which water is dispersed phase and fat is dispersion medium.
Cream.
Q. 86.Define emulsifying agent.
Ans.Substance which is added to the emulsion for its stabilization is called emulsifying agent.
Examples .
For O/W emulsions:
Proteins, gums, natural and synthetic soaps, etc.,
For W/O emulsions :
Heavy metal salts of fatty acids, long chain alcohols, lampblack, etc.
Q.87.Explain the methods for separation of dispersed phase from dispersion medium in emulsion.
Ans.
- Heating,
- Freezing,
- Centrifuging
Q.88 The color of the sky is blue why.?
Ans. It is due to scattering of blue light by dust particles along with water suspended in air.
Q.89.What ccauses fog and mist.
Ans.The air above the ground level cooled below its dew point ,causing water vapor to condenses around the dust and other particle in the atmosphere and results in formation of fog and mist.
Q.90.What causes rain.
Ans.1.When cloud droplets in clouds grow and combine to become so large till they come down in the form of rain.
2.Rain fall also occurs due to precipitation when two oppositely charged clouds meet ,they neutralises each other and come down in the form of rain.
Q.91.How artificial rain is possible.
Ans.Artificial rains are possible by throwing electrified sand to the colloid from aeroplanes which convert water to rain fall.
Q.92.Give some examples of food articles of which are colloidal in nature.
Ans. Milk, butter, halwa, ice creams, and fruit juices.
Q.93.Why alum has styptic properties and stop further bleeding caused by razor shaving.
Ans.The styptic action of alum due to coagulation of blood forming a clot which stops further bleeding.
Q.94.How formation of delta occurs ?
Ans.It is an example of coagulation .River water is a colloidal solution of clay. When river water meets the sea water, the electrolytes present in sea water coagulate the colloidal solution of clay and delta forms.
Q.95 What is Cottrell precipitator.
Ans.Cottrell precipitator is precipitator which precipitate smoke particles (such as carbon, arsenic compounds, dust) because this precipitator contains plates having a charge opposite to that carried by smoke particles.
Q96.How alum make water fit for drinking.
Ans. Alum is used as flocculating ion and coagulate the suspended impurities in water and make water fit for drinking purposes.
Q.97.Give some examples of medicine which are colloids.
Ans.
- Argyrol (silver sol ) is used as an eye lotion.
- Colloidal antimony is used in curing kalaazar.
- Colloidal gold is used for intramuscular injection.
- Milk of (emulsion) is used for stomach disorders.
Q.98.Colloidal medicines are more effective medicines why?
Ans.Due to their large surface area colloidal medicines can be easily assimilated thus colloidal medicines are more effective medicines.
Q.99.Explain the tanning process.
Ans.The process by which animal hide which is a +ve charged colloid is made hard by soaking it tannin which is –ve charged colloid.
Q.100.What is latex.
Ans.Latex is the emulsion of polymer microparticles in an aqueous medium.
Q.101.How can we obtained rubber from latex.
Ans. Rubber is obtained by coagulation of latex.
Q.102.Give examples of some Industrial products which are colloidal in nature.
Ans.Paints, inks, synthetic plastics, rubber,graphite lubricants, cement etc.
