The p-block elements
Q.1.Group number 13-18 elements are known as p-block elements why?
Ans.The last electron of these elements goes into p-orbitals.
Q.2.What is the valance shell electronic configuration of p-block elements.
Ans.ns2np1-6
Q.3.1st member of group 13-17 elements show different behavior from rest of the elements why?
Ans.It is due to small size and high electronegativity of 1st member of group 13-17 elements.
Q.4.1st member of group 13-17 elements can show maximum covalency four but other elements can expand their covalency greater than four why?
Or
While boron forms only [BF4]–, aluminium gives [AlF6]3– ion why?
Ans.1st member of group 13-17 elements has only four outermost orbitals one ns and three np orbitals hence they can show maximum covalency four but other elements have vacant d orbitals hence hence they can expand their covalency greater than four.
Q.5.The first member of group 13-17 elements can form multiple bonds to itself ( e.g., C=C, C≡C, N≡N) and to other elements like C=O, C=N, C ≡N, N=O). But heavier member don’t why?
Ans.It is due to small size and high electronegativity of 1st member of group 13-17 elements but p-orbital of heavier member is so large and diffuse to effective overlapping.
Q.6.What is inert pair effect?
Ans.When 2 electrons of outermost ns orbital are reluctant towards bonding and elements show oxidation state two unit less than the group oxidation state ,this effect is called inert pair effect.
Group 13 elements :boron family
B,Al,Ga,In, Tl
Q.1.Boron has unusually high melting point why?
Ans.Due to very strong crystalline lattice arises from its small size, boron has unusually high melting point.
Q.2. Gallium can exist in liquid state during summer why?
Ans. It is due to very low melting point of Gallium.
Q.3.Boron forms only covalent compound why?
Ans.It is due to very high first three ionization enthalpies of boron it is unable to form +3 ions hence it forms mainly covalent compound.
Q.4.Aluminium is electropositive element and can form ionic compound why?
Ans.It is due to larger size and lower ionization enthalpies of Al can form Al3+ion and can form ionic compound.
Q.5.What are the oxidation states of group 13 elements with or without inert pair effect.
Ans.Without inert pair effect +3
With inert pair effect +1
Q.6.Stability of +3 oxidation state decreases top to bottom and +1 increases top to bottom.
Ans .As we go down the group electronegativity decreases hence energy required to unpair two ns electron is not compensated by energy release in formation of two extra bond.
Q.7.Which is more stable in BCl3 and TlCl3.
Ans.Since stability of +3 oxidation stability decreases top to bottom as energy required to unpair two ns electron is not compensated by energy release in formation of two extra bonds hence the BCl3 is more stable than TlCl3.
Q.8.Atomic radius of Ga is less than that of Al why?.
Ans. Presence of additional 10 d-electrons in Ga which produce poor screening effect and increased effective nuclear charge decreases the atomic size.
Q.9.Ionisation enthalpy of Ga is higher than Al why?
Ans. Atomic size of Ga is lower than Al hence Ionisation enthalpy of Ga is higher than Al
Q.10.Ionisation enthalpy of Tl is higher than In why.
Ans Higher ionization energy compensate the high effective nuclear charge arises due to poor shielding effect of d and f electrons in Tl.
Q.11.The order of ionisation enthalpies of group 13 elements increases in following order why ?
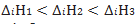
Ans. As the elements losing its electron +ve charge increases thus size decreases hence ionization enthalpy increases.
Q.12.Electronegativity of alkali metals first decreases from B and Al and then increases slightly why?
Ans.It is due to irregularities in atomic size and presence of d and f electrons in heavier member.
Q.13.In trivalent state compounds of 13 elements are Lewis acid why?
Ans. In trivalent state compounds of 13 elements are electron deficient hence they are Lewis acid example BF3, BCl3 etc.
Q.14. Arrange the Lewis basic strength of group 13 trivalent compound.
Ans. The tendency to behave as Lewis acid decreases with the increase in the size down the group. BI3< BBr3< BCl3< BF3
Q.15. Aluminum is very less reactive towards oxygen why?
Ans. Aluminum forms a thin oxide layer on the surface which protects the metal from further attack.
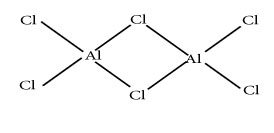
Q.16.AlCl3 exists in dimer why?
Ans. Al atom in molecule is electron deficient due to presence of only six electrons on its valence shell.
Q.17.Arrange acidic property of group 13 oxides.
Ans.Acidic property of group 13 oxide decreases down the group.
Boron trioxide – acidic
Aluminum and gallium oxides – amphoteric
Indium and thallium – basic
Q.18.White fumes appear around the bottle of anhydrous aluminum chloride give reason.
Ans.Anhydrous aluminum chloride liberates HCl gas on hydrolysis with atmospheric moisture moist HCl appears white in colour.
Q.19.Boron is unable to form BF63-why?
Ans. Boron has only four outermost orbital one ns and three np orbitals hence it cannot expand covalency greater than 4.
Q.20.What is the heating effect of borax?
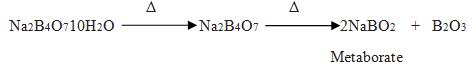
Q.21.What is borax bead test?
Ans.This test is used for identify transition elements. When borax is converted into metaborate of a transition elements it gives characteristic color.
Q.22.The structure of boric acid H3BO3.
Ans .It has a layer structure in which planar BO3 units are joined by hydrogen bonds.
Q.23.What is the heating effect of boric acid?
Ans.
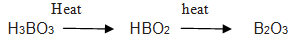
Q.24.Boric acid is weak monobasic acid why?
Ans.Boric acid accept OH– from water in turn release H+ ion, thus orthoboric acid behaves like monobasic acid.
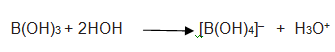
Q.25.What is inorganic benzene
Ans .B3N3H6 is known as inorganic benzene.
Q.26.Describe the structure of diborane.
Ans. Diborane contains four terminal B-H bonds and two bridge (B-H-B) bonds .The four terminal B-H bonds lie in one plane and two centre-two electron bonds bonds while the two bridge (B-H-B) bonds present above and below this plane and three centre–two electron bonds.
Q.27.What do you mean by banana bond in diborane.
Ans. Two bridge (B-H-B) bonds present above and below this plane and three centre–two electron bonds are known as banana bond.
Q.28.What are the different uses of B and Al.
Ans.1.Boron fibers in making bullet proof vest.
2 Metal borides are in nuclear industry.
3.Borax and boric acid are used in making heat resistant glasses.
4. Aq. orthoboric acid as mild antiseptic.
5. In packing
6. Utensil making,
7.Construction, aeroplane and transportation industry.
Group 14 elements: Carbon family
C,Si,Ge,Sn,Pb
Q.30.What is the common oxidation state of group 14 elements?
Ans. +4 and +2 are the common oxidation states of these elements. Carbon also show negative oxidation state.
Q.31.Compounds in +4 oxidation state are generally covalent in nature why?
Ans. Since the sum of the first four ionization enthalpies of group 14 elements is very high hence these compounds are mainly covalent.
Q.32.What are the oxidation state of group 14 elements without inert pair effect.
Ans +2=With inert pair effect
+4=without inert pair effect
Q.33.Stability of +2 oxidation state increases top to bottom and +4 decreases top to bottom why?
Or
PbX2 is more stable than PbX4 why?
Or
GeX4 is more stable than GeX2
Or
In heavier members of group 14 elements the tendency to show +2 oxidation state increases in the sequence Ge<Sn<Pb.why?
Or
Carbon and silicon mostly show +4 oxidation state why?
Ans. As we go down the group electronegativity decreases hence energy required to unpair two ns electrons is not compensated by energy release in formation of two extra bond.Thus stability of+2 oxidation stae increases and that of +4 decreases.
Q.34.Lead compound in +4 state are strong oxidising agents why?
Ans. +4 oxidation state of Pb is unstable due to inert pair effect hence tend to decrease this oxidation state into stable +2 oxidation state hence works as oxidizing agent.
Q.35. Sn in +2 state is a reducing agent why?
Ans.Sn can show both +2 and +4 oxidation state hence in +2 oxidation state it works as reducing agent.
Q.36.Maximum covalency of carbon is 4 but other elements can extend their covalency greater than 4 why?
Or
The species like, SiF62–, [GeCl6]2–,[Sn(OH)6]2- of heavier group 14 elements known but carbon is unable to form these types of compound why?
Or
Halides of heaviour elements(SiCl4) undergo hydrolysis but CCl4 not why?
Ans. Carbon has only four outermost orbitals one 2s and three 2p orbitals hence they can show maximum covalency four but other elements have vacant d orbitals hence hence they can expand their covalency greater than four.
Q.37.Give the example of acidic,amphoteric and neutral oxide of group14 elements
Ans. CO2, SiO2 and GeO2 –acidic
SnO2 and PbO2 – amphoteric SnO PbO
CO –neutral
Q.38.PbI4 does not exist why?
Ans. Energy released during formation of Pb—I bond cannot unpair 6s2 electrons and excite of them to higher orbital to have four unpaired electrons around lead atom.
Q.39.[SiF6]2– is known whereas [SiCl6]2– not why?
Ans.(i) Due to larger size of Cl– ions they cannot be accommodated around Si4+
(ii)Due to lower electronegativity of Cl– Interaction between lone pair of chloride ion and Si4+ is not very strong.
Q.40.Carbon show different behaviour than rest of the members of its Group why?
Ans. It is due to following reasons of carbon atoms.
Smaller size
High electronegativity
Higher ionisation enthalpy
Unavailability of d orbitals
Q.41.What is catenation property.
Ans.Tendency of atoms to link with one another through covalent bonds to form chains and rings called catenation.
Q.42.Catenation property of carbon is very high why?
Ans.This is due to strong C—C bond catenation property of carbon is very high.
Q.43.Tendency to show catenation property by group 14 elements decreases down the group why?
Ans.The size and electronegativity decreases down the group hence tendency to show catenation property by group 14 elements decreases down the group. Lead does not show catenation. C > > Si > Ge = Sn.
Q.44.Carbon is able to show allotropic forms why?
Ans.Due to high catenation property and ability to form![]() bond by carbon atoms it show allotrope.
bond by carbon atoms it show allotrope.
Q.45.Name the all crystalline allotropes of carbon.
Ans.(i)Diamond
(ii)Graphite
(iii) Fullerenes
Q.46.Describe the structure of Diamond.
Ans. Diamond has crystalline structure in which each carbon is sp3 hybridised and linked to four other carbon in tetrahedral manner.
Q.47.Diamond is used as an abrasive for sharpening hard tools why?
Or
Diamond is covalent, yet it has high melting point why?
Ans.Diamond is a hardest substance on the earth because bond dissociation enthalpy of extended covalent bonding between carbon atoms is very high hence use as abrasive.
Q.48.What are the other uses of diamond.
Ans.(i) In jewelry
(ii) In making dyes
(ii) In the manufacture of tungsten filaments for electric light bulbs.
Q.49.Describe the structure of Graphite.
Ans (i) Crystalline layered structure.
(ii) Each layer is joined with each other by van der Waals forces.
(iii) Each layer is composed of planar hexagonal rings of carbon atoms.
(iv)Each carbon is sp2 hybridised in hexagonal ring.
(v) Fourth electrons of carbon are delocalized over the whole sheet.
Q.50.Graphite conducts electricity why?
Ans. Since each carbon is sp2 hybridised in hexagonal ring hence fourth electrons of carbon are delocalized over the whole sheet thus graphite is good conductor.
Q.51.Graphite is very soft and slippery why?
Or
Graphite is used as a dry lubricant in machines running at high temperature, where oil cannot be used as a lubricant.
Ans.Due to layered structure graphite cleaves easily between the layers and, therefore it is very soft and slippery.
Q.52.How is fullerene made?
Ans.By heating graphite in an electric arc in the presence of inert gases.
Q.53.Describe the structure of Fullerenes.
Ans. Fullerene contains mainly C60 molecule with traces of C70.
Q.54.Describe the structure of C60 molecule(Buckminsterfullerene)
Ans.(i) It contains 20 six- membered rings and 12 five membered rings.
(ii) A six membered ring is fused with six or five membered rings but a five membered ring can only fuse with six membered rings.
(iii)All the carbon atoms are sp2 hybridised
(iv) The fourth electron of each carbon is delocalised in molecular orbitals.
Q.55.C60 molecule is also called Buckminsterfullerene why?
Ans. C60 molecule has a shape like soccer ball and called Buckminsterfullerene.
Q.56.Which Property of Buckminsterfullerene give its aromatic character?
Ans. The fourth electron of each carbon is delocalised in molecular orbitals.
Q.57.Fullerenes are the only pure form of carbon why?
Ans Fullerenes have smooth structure without having ‘dangling’ bonds.
Q.58. ![]() of graphite is taken as zero why?
of graphite is taken as zero why?
Ans. Graphite is thermodynamically most stable allotrope of carbon and, therefore ![]() of graphite is taken as zero.
of graphite is taken as zero.
Q.59.Give some examples of impure form of carbon.
Ans. Carbon black, coke, and charcoal.
Q.60.How is Carbon black obtained?
Ans. Carbon black is obtained by burning hydrocarbons in a limited supply of air.
Q.61.How is charcoal obtained?
Ans.Charcoal is obtained by heating wood in the absence of air.
Q.62 How is coke obtained?
Ans. Coke is obtained by heating coal in the absence of air.
Q.63.What are the uses of carbon?
Ans.(i) Composites which contain graphite are used in products such as tennis rackets, fishing rods, aircrafts and canoes.
(ii) Graphite is used as electrode in batteries and industrial electrolysis due to its good conducting nature.
(iii) Charcoal is used in gas masks to adsorb poisonous gases due to its porous nature.
(iv) Carbon black is used as black pigment in black ink and as filler in automobile tyres.
(v) Coke is used as a fuel and largely as a reducing agent in metallurgy.
(vi) Diamond in jewellery.
Q.64.What is producer gas?
Ans.Mixture of CO and N2 known as producer gas.
Q.65.What is water gas/synthesis gas/syn gas?
Ans.Mixture of CO and H2 known as producer gas.
Q.66.What are metal carbonyls?
Ans.CO contains a lone pair at carbon and acts as donor atom to the metal and forms metal carbonyl.
Q.67.CO is highly poisonous why?
Ans.CO forms complex with hemoglobin which is more stable than the oxygen-hemoglobin complex prevents hemoglobin in the red blood corpuscles from carrying oxygen round the body and ultimately death.
Q.68.Name the buffer system which helps blood to maintain pH.
Ans.H2CO3/HCO3–
Q.69.What is photosynthesis?
Ans.The process by which green plants convert atmospheric CO2 into carbohydrates called photosynthesis.
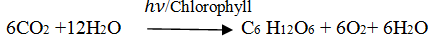
Q.70.What is green house effect?
Ans. Increasing amount of CO2 in atmosphere produce during combustion of fossil fuels and decomposition of limestone for cement raise the temperature of the atmosphere called green house effect.
Q.71.What is dry ice?
Ans.Solid CO2 is known as dry ice. Dry ice is used as a refrigerant for ice-cream and frozen food.
Q.72. What are the uses of gaseous CO2?
Ans (i)As and non-supporter of combustion it is used as fire extinguisher.
(ii) In soft drinks.
Q.73.Draw the resonating structure of CO2
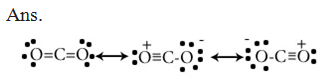
Q.74 What is silica?
Ans.Silicon dioxide(SiO2) is known as silica. Example Quartz, cristobalite and tridymite are crystalline forms of silica.
Q.75.Describe the structure of silica.
Ans.Silicon dioxide is a covalent, three-dimensional network solid in which each silicon atom is covalently bonded in a tetrahedral manner to four oxygen atoms.
Q.76.Silica in its normal form is almost nonreactive why?
Ans. It is due to very high Si—O bond enthalpy.
Q.77.What are the different uses of silica?
Ans (i) Quartz is used as a piezoelectric material
(ii) Silica gel is used as a drying agent.
(iii) Kieselghur, an amorphous form of silica is used in filtration plants.
Q.78.What are silicones ?
Ans.silicones are organosilicon polymers formed by –(R2SiO)-as repeated unit.
Q.79.What are the uses of silicones?
Ans. Silicones are used as sealant, greases, electrical insulators and for water proofing of
fabrics.
(ii) In surgical and cosmetic plants.
Q80 What are silicates?
Ans.The basic structural unit of silicates is SiO4 in which silicon atom is bonded to four oxygen atoms in tetrahedron manner.Example feldspar, zeolites, mica and asbestos.
Q81.Give the two examples of man made silicate.
Ans.Glass and cement.
Q82 What are Zeolites?
Ans. If some Aluminum atoms replace few silicon atoms in three-dimensional network of silicon dioxide the structure formed is aluminosilicate a types of zeolite.
Q 83.What are different uses of zeolites?
Ans.(i)ZSM-5 a zeolite used in used to convert alcohols directly into gasoline.
(ii)Hydrated zeolites are used for softening of “hard” water
